Related Subjects:
|AP of the Bone Marrow
|AP of the Spleen
|AP of the Thymus Gland
🫁🧠 The thymus is a primary lymphoid organ responsible for T-cell development (“education”) and central immune tolerance.
It is largest and most active in childhood, then gradually involutes after puberty (functional tissue replaced by fat), but it remains immunologically relevant.
Clinically, the thymus matters because errors in thymic selection underpin autoimmunity, and thymic disease is strongly associated with myasthenia gravis.
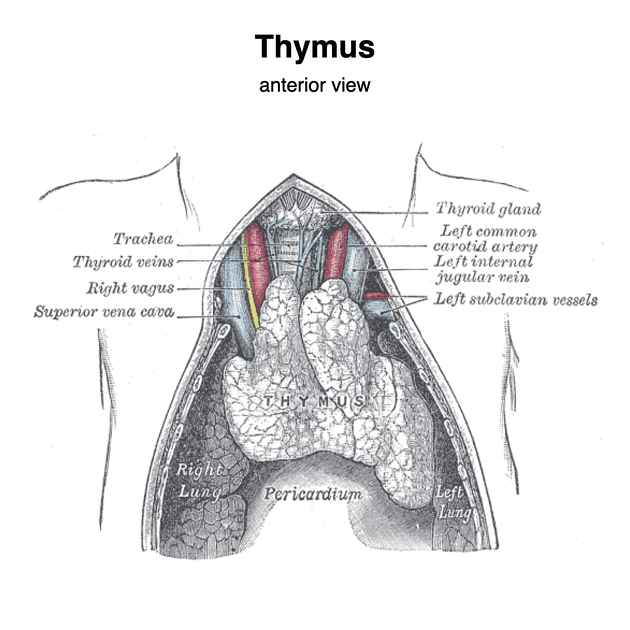
📍 1) Gross Anatomy and Relations
- Location: Anterior mediastinum (superior + anterior part), behind the sternum and in front of the pericardium/great vessels.
- Extent: Often extends from the lower neck (beneath thyroid) into the upper anterior mediastinum, especially in children.
- Shape: Bilobed, soft gland (two asymmetric lobes joined by connective tissue).
- Key relations (high yield):
- Anterior: Sternum
- Posterior: Pericardium, great vessels (SVC, brachiocephalic veins, aortic arch branches), trachea
- Lateral: Pleura and lungs
📌 Clinical: In children, a prominent thymic shadow can look dramatic on CXR but may be normal. In adults, an anterior mediastinal mass includes the classic “4 Ts”:
Thymoma, Teratoma, Thyroid (ectopic/goitre), Terrible lymphoma.
🧱 2) Microscopic Anatomy (How It’s Built)
- Capsule and septa: A connective tissue capsule sends septa inward, dividing the thymus into lobules.
- Cortex (outer, darker): Densely packed immature thymocytes (developing T cells) + thymic epithelial cells.
- Medulla (inner, paler): Fewer lymphocytes; enriched in thymic epithelial cells, dendritic cells, macrophages.
- Hassall’s corpuscles: Whorls of epithelial cells in the medulla (characteristic thymic histology); involved in T-cell maturation and tolerance signalling.
- Unique feature: The thymus has an effective blood–thymus barrier (especially in cortex), reducing antigen exposure during early T-cell development.
🩸 3) Blood Supply, Venous Drainage, Lymphatics, Innervation
- Arterial supply: Predominantly from internal thoracic arteries and branches of the inferior thyroid artery (variable).
- Venous drainage: To left brachiocephalic vein and internal thoracic veins (variable).
- Lymphatics:
- Thymus has efferent lymphatics (drain out) but essentially no afferent lymphatics (little incoming antigen sampling).
- Innervation: Autonomic fibres (sympathetic and parasympathetic) modulate stromal/vascular tone; immune “education” is mainly local cytokine-driven.
🧪 4) Physiology: What the Thymus Does
🌱 A) T-cell Development (Thymopoiesis)
T cells originate from bone marrow stem cells but mature in the thymus. The thymus provides a specialised microenvironment (“niche”) that
drives T-cell receptor (TCR) gene rearrangement, selection, and lineage commitment into CD4 or CD8 T cells.
- Entry: Bone-marrow derived precursors enter via blood at the corticomedullary junction.
- Early stages (cortex): Cells rearrange TCR genes; become “double positive” (CD4+ and CD8+) thymocytes.
- Positive selection (cortex): Thymic epithelial cells present self-peptides on MHC.
- Cells must recognise self-MHC weakly/moderately to survive (otherwise they die by neglect).
- This “teaches” T cells to be MHC-restricted (they can only respond when antigen is presented on your own MHC).
- Lineage commitment:
- Recognition of MHC II → becomes CD4+ helper T cell.
- Recognition of MHC I → becomes CD8+ cytotoxic T cell.
🛡️ B) Central Tolerance (Preventing Autoimmunity)
Central tolerance is the thymus’s “quality control”: it eliminates strongly self-reactive T cells and generates regulatory populations.
Failure here increases the risk of autoimmune disease.
- Negative selection (mainly medulla): Dendritic cells and medullary thymic epithelial cells present self-antigens.
Strongly self-reactive thymocytes undergo apoptosis.
- AIRE gene (Autoimmune Regulator): Drives expression of tissue-restricted antigens in the thymus (e.g., pancreatic, thyroid proteins),
allowing deletion of T cells that would attack peripheral organs.
- Treg generation: Some self-reactive cells are diverted into regulatory T cells (Tregs) (e.g., FoxP3+), which suppress autoimmunity in the periphery.
🧫 5) Thymic Hormones and Signalling Molecules
- Thymosin, thymopoietin, thymulin: Historical “thymic hormones” that support T-cell differentiation and maturation signalling.
- Cytokine environment: Local IL-7 and stromal signals are crucial for thymocyte survival and proliferation.
🧠 In practice, think of the thymus less as a classic endocrine gland and more as an immune training academy using epithelial scaffolding + cytokines to shape T-cells.
📈 6) Age-Related Changes (Involution)
- Childhood: Large, highly active thymus; high output of naïve T cells.
- Puberty onward: Progressive involution with fatty replacement; thymic output declines.
- Adult life: Peripheral T-cell homeostasis relies more on memory T-cell expansion, but thymic function does not drop to zero.
⚠️ 7) Clinical Relevance
🎗️ A) Thymoma and Myasthenia Gravis
- Thymoma: Tumour of thymic epithelial cells; may be asymptomatic or present as anterior mediastinal mass.
- Association: Strong link with myasthenia gravis (autoantibodies against ACh receptors or related proteins).
- Other autoimmunity: Thymoma can be associated with other immune dysregulation syndromes (paraneoplastic).
🧬 B) DiGeorge Syndrome (22q11 deletion)
- Mechanism: Failed development of 3rd/4th pharyngeal pouches → thymic hypoplasia/aplasia.
- Immune consequence: Low/absent T cells → recurrent viral/fungal infections.
- Classic triad: Thymic aplasia, hypocalcaemia (parathyroid hypoplasia), conotruncal cardiac defects.
🦠 C) Immunodeficiency Patterns
- Thymic dysfunction tends to impair cell-mediated immunity (viral, fungal, opportunistic infections).
- Secondary effects include weaker B-cell help → reduced class switching and antibody quality.
🩻 D) Imaging Pearl
- Normal thymus in children can appear prominent; “sail sign” may be a normal variant.
- Thymic rebound hyperplasia can occur after stress (e.g., chemotherapy, severe infection) and may mimic mass.
📊 Quick Revision Table
| Feature |
Key Point |
Clinical Link |
| Location |
Anterior mediastinum; prominent in childhood |
Anterior mediastinal masses (“4 Ts”) |
| Structure |
Lobules with cortex (dense thymocytes) and medulla (Hassall’s corpuscles) |
Histology clue for thymic tissue |
| Main function |
T-cell maturation + central tolerance |
Autoimmunity if selection fails |
| Positive selection |
Survival if weak/moderate recognition of self-MHC |
MHC restriction |
| Negative selection |
Deletion of strongly self-reactive cells (AIRE helps present tissue antigens) |
Prevents autoimmune disease |
| Involution |
Fatty replacement after puberty; reduced naïve T-cell output |
Age-related immune changes |
📝 Summary
The thymus is the body’s T-cell “training centre”: it shapes developing thymocytes into functional CD4 and CD8 T cells that recognise antigen on
self-MHC while eliminating dangerous self-reactive clones through central tolerance (including AIRE-mediated antigen display).
Although it involutes after puberty, its early-life role is critical for establishing a competent and self-tolerant adaptive immune system.
Clinically, remember the thymus in myasthenia gravis/thymoma and in T-cell immunodeficiency syndromes such as DiGeorge.

