| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Subdural haematoma
Related Subjects: |Brain Herniation syndromes |Epidural Haematoma |Subdural haematoma |Extradural haematoma🩸 A Subdural Haematoma (SDH) is bleeding into the potential space between the dura mater and arachnoid mater. 🧠 Classically venous in origin (torn bridging veins), producing a slower and often insidious presentation compared with extradural haematoma. ⚠️ Always suspect SDH in elderly, alcohol-dependent, anticoagulated, or cognitively declining patients after even minor trauma.
💡 Pathophysiology (What is really happening)
- Acceleration–deceleration forces tear bridging veins traversing from cortex to dural sinuses.
- Venous pressure is low → gradual accumulation → delayed neurological decline ⏳.
- Cerebral atrophy (ageing, alcohol misuse) increases vein length and fragility.
- In chronic SDH, inflammatory membranes form and become vascularised (often via middle meningeal artery branches), predisposing to recurrence.
- Mass effect → midline shift → raised intracranial pressure (ICP) and risk of herniation.
🧬 Classification by Time Course
- Acute: <72 hours (hyperdense on CT).
- Subacute: 3–21 days (may appear isodense).
- Chronic: >3 weeks (hypodense; liquefied).
📌 Risk Factors
- Falls (especially >65 years) 👴
- Alcohol misuse 🍺
- Anticoagulants (warfarin, DOACs) and antiplatelets 💊
- Coagulopathy or thrombocytopenia
- Low CSF pressure (post-LP, VP shunt)
- Epilepsy or high-impact trauma 🚗
🩺 Clinical Features
Presentation varies with bleed rate and cerebral reserve. Acute SDH often mimics severe TBI; chronic SDH frequently masquerades as delirium or dementia.
- Acute: ↓ GCS, severe headache 🤕, vomiting, seizures, focal deficit.
- Chronic: Progressive confusion, personality change, gait disturbance, falls.
- Focal signs: hemiparesis, aphasia, visual field deficit.
- Unilateral dilated pupil (CN III compression) → impending herniation 🚨.
- Cushing response: bradycardia + hypertension + irregular breathing.
📷 Imaging
CT Head (First-line)
- Crescent-shaped extra-axial collection.
- Crosses suture lines but limited by dural reflections (falx, tentorium).
- Assess thickness, midline shift, basal cistern effacement.
Large Subacute SDH with Midline Shift
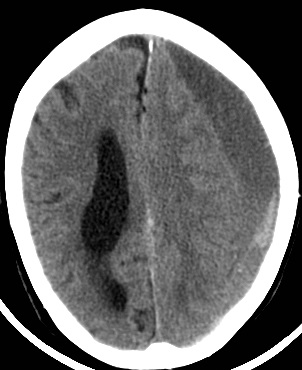
Large Acute SDH
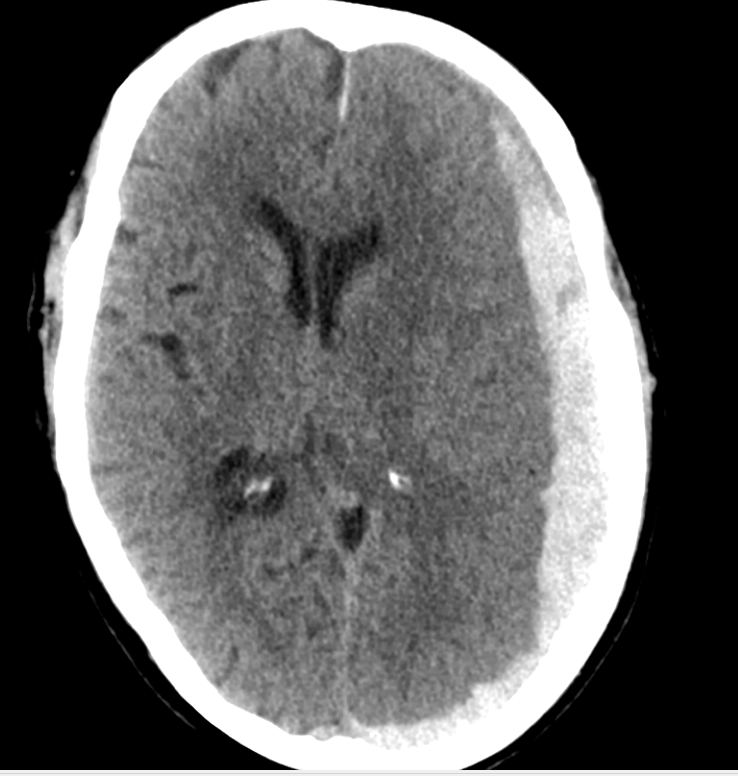
MRI is helpful when CT is equivocal (isodense collections, posterior fossa, chronic cases).
🔬 Investigations
- Bloods: FBC, U&E, clotting screen, group & save.
- Urgently check INR if on warfarin.
- Platelets ideally >100 × 10⁹/L for surgery.
- CT cervical spine in trauma.
⚠️ Complications
- Raised ICP and transtentorial herniation 🧠
- Recurrent chronic SDH (10–20%)
- Seizures
- Infarction from vascular compression
- Subdural empyema (rare)
⚡ Management
All moderate or large SDHs require early neurosurgical discussion. Management is guided by neurological status and imaging findings.
- Initial (ABC): Airway protection if GCS ≤8; maintain normoxia and normocapnia.
- Reverse anticoagulation:
- Warfarin → IV vitamin K + PCC.
- DOAC → specific reversal agents if available.
- Surgical Indications:
- Thickness >10 mm OR midline shift >5 mm.
- Neurological deterioration.
- Acute SDH: Craniotomy ± decompressive craniectomy.
- Chronic SDH: Burr-hole drainage ± subdural drain.
- Conservative: Small, asymptomatic SDH with close observation and repeat imaging 👀.
- Consider MMA embolization in recurrent chronic SDH.
🆘 Red Flags (Call Neurosurgery Immediately)
- Deteriorating GCS
- New pupillary asymmetry
- Progressive focal deficit
- Significant midline shift on imaging
📋 EDH vs SDH Comparison
🟥 EDH: Arterial (middle meningeal), biconvex, does NOT cross sutures, lucid interval possible.
🟦 SDH: Venous, crescent-shaped, CAN cross sutures, often delayed presentation.
⚠️ Confusion + fall + anticoagulation = SDH until proven otherwise.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
