| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Microtubules
Related Subjects: |DNA replication |DNA structure in Nucleus |Mitosis and Meiosis |Ribosomes |Microtubules |Mitochondria |Smooth and Rough Endoplasmic Reticulum
🧬 Microtubules are highly organised, GTP-dependent polymers of tubulin that form the largest and most dynamic component of the cytoskeleton. They act as mechanical scaffolds, intracellular transport highways, and force-generating elements for mitosis and motility. In clinical medicine, microtubule dysfunction explains key features of neurodegeneration, cancer progression, ciliopathies, and drug toxicity.
🧱 Molecular Architecture and Biogenesis
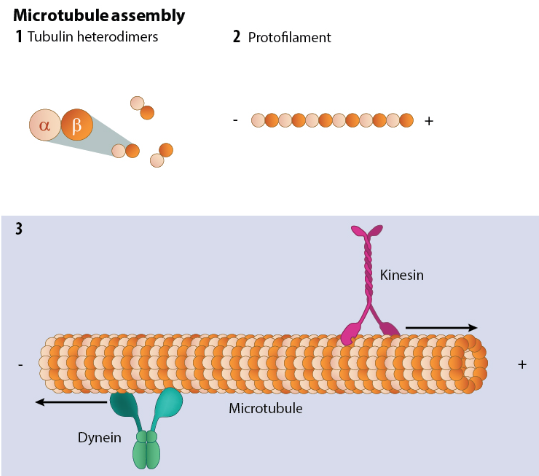
- Tubulin Subunits
- Composed of α-tubulin and β-tubulin heterodimers.
- Both bind GTP, but only β-tubulin hydrolyses it.
- GTP hydrolysis alters tubulin conformation → instability.
- Multiple tubulin isoforms exist → tissue-specific properties.
- Protofilaments and Tubular Assembly
- Dimers assemble head-to-tail → linear protofilaments.
- Typically 13 protofilaments align laterally.
- Form a hollow cylinder (~25 nm diameter).
- Lateral bonding gives mechanical rigidity.
- Nucleation and the MTOC
- Microtubules rarely self-assemble spontaneously.
- Initiated at microtubule-organising centres (MTOCs).
- Centrosomes contain γ-tubulin ring complexes.
- These act as “templates” for polymerisation.
➕➖ Polarity, Orientation, and Cellular Geometry
- Microtubules are intrinsically polar structures.
- Plus (+) end:
- Fast-growing, unstable.
- Faces cell periphery.
- Main site of remodelling.
- Minus (−) end:
- More stable.
- Anchored to centrosome.
- Polarity determines intracellular traffic direction.
- Establishes cell polarity (e.g. neuronal axons).
⚙️ Dynamic Instability and GTP Cap Regulation
- Microtubules constantly alternate between states:
- Polymerisation (growth)
- Catastrophe (rapid shrinkage)
- Rescue (recovery)
- Stability depends on the GTP cap.
- GTP-tubulin stabilises the plus end.
- Loss of GTP cap → protofilament curling → collapse.
- Enables rapid network reorganisation.
- Essential for mitosis, synapse formation, and migration.
🧠 Teaching point: Dynamic instability allows cells to “search and capture” chromosomes during mitosis. Without this, spindle assembly fails and apoptosis follows.
🔬 Functional Roles in Cellular Physiology
- Mechanical Support and Cell Shape
- Resist compressive forces.
- Maintain elongated morphology in neurons.
- Support epithelial polarity.
- Intracellular Transport Networks
- Serve as bidirectional transport tracks.
- Enable long-distance trafficking.
- Transport:
- Mitochondria → energy distribution
- Synaptic vesicles → neurotransmission
- Lysosomes → autophagy
- mRNA → local protein synthesis
- Failure → “dying-back” neuropathy.
- Cell Cycle Control
- Form bipolar mitotic spindle.
- Attach to kinetochores.
- Generate pulling forces.
- Ensure accurate chromosomal segregation.
- Defects → aneuploidy and malignancy.
- Ciliary and Flagellar Motility
- Axoneme = 9 peripheral + 2 central microtubules.
- Dynein converts ATP to mechanical force.
- Generates wave-like motion.
- Clears mucus and moves gametes.
- Organelle Positioning
- Determine location of Golgi apparatus.
- Organise endoplasmic reticulum.
- Maintain nuclear position.
🧩 Microtubule-Associated Proteins (MAPs): Regulation and Control
- Structural MAPs
- Tau, MAP2, MAP4.
- Cross-link microtubules.
- Increase rigidity.
- Prominent in CNS.
- Destabilising Proteins
- Stathmin: sequesters tubulin dimers.
- Katanin: severs filaments.
- Spastin: remodels networks.
- Motor MAPs
- Kinesin families: anterograde transport.
- Dynein-dynactin: retrograde transport.
- Critical for synaptic maintenance.
🧬 Integration with Cellular Metabolism and Signalling
- Highly ATP-dependent system.
- Coupled to mitochondrial distribution.
- Regulated by phosphorylation pathways.
- Respond to growth factors (EGF, NGF).
- Modulated by intracellular calcium.
- Interact with actin for cell migration.
🏥 Clinical Relevance and Pathophysiology
- Neurodegeneration
- Tau hyperphosphorylation → detachment.
- Microtubule collapse.
- Transport failure.
- Synaptic loss → cognitive decline.
- Peripheral Neuropathy
- Seen in diabetes, toxins, chemotherapy.
- Due to axonal transport failure.
- Cancer Biology
- Rapid proliferation requires intact spindles.
- Microtubule disruption → mitotic catastrophe.
- Resistance involves tubulin mutations.
- Ciliopathies
- Defective axonemal microtubules.
- Lead to:
- Chronic sinusitis
- Bronchiectasis
- Infertility
- Situs inversus
- Renal cysts
- Neurotropic Infections
- Viruses use dynein to reach CNS.
- Explains latency and reactivation.
💊 Pharmacology: Drugs Targeting Microtubules
- Colchicine
- Blocks tubulin polymerisation.
- Inhibits neutrophil migration.
- Used in gout and pericarditis.
- Vinca Alkaloids
- Prevent assembly.
- Cause metaphase arrest.
- Side effect: peripheral neuropathy.
- Taxanes
- Stabilise microtubules.
- Prevent disassembly.
- Cause myelosuppression.
- Anti-parasitic / Antifungal Agents
- Mebendazole, griseofulvin.
- Exploit cytoskeletal dependence.
🧪 Diagnostic and Laboratory Clues
- Peripheral neuropathy during chemotherapy.
- Recurrent chest infections + infertility.
- Early dementia with movement disorder.
- Abnormal sperm motility.
🧠 High-Yield Exam Pearls
- 13 protofilaments = classic structure.
- Plus end = dynamic.
- Minus end = centrosomal.
- Dynamic instability = mitosis.
- Tau stabilises microtubules.
- Spindle drugs → neuropathy.
📌 Summary
Microtubules are polar, GTP-regulated polymers that coordinate intracellular transport, cell division, and motility. Their behaviour is governed by dynamic instability and tightly regulated by specialised proteins. Failure of microtubule systems underlies major neurological, oncological, respiratory, and metabolic diseases, making them fundamental to clinical and translational medicine.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
