| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Clotting pathways
🩸 Haemostasis and Coagulation
- Haemostasis means the arrest of bleeding after vascular injury.
- The body first forms a primary platelet plug, but this alone is relatively fragile and can break down.
- The coagulation cascade then generates fibrin, which reinforces and stabilises the platelet plug, reducing the risk of further bleeding.
- Effective haemostasis therefore depends on three closely linked components:
- the blood vessel wall
- platelets
- the coagulation system
🫀 The Vascular Phase
- Blood vessels must maintain blood flow under normal conditions, but also respond rapidly to injury.
- The innermost layer is the endothelium, a single layer of endothelial cells lying on a basement membrane.
- Endothelial cells produce substances that normally make the vessel lining antithrombotic, including:
- prostacyclin (PGI₂), which inhibits platelet aggregation
- nitric oxide (NO), which promotes vasodilation and inhibits platelet activation
- ecto-ADPase, which degrades ADP released by platelets
- heparan sulphate and antithrombin-mediated anticoagulant activity
- tissue plasminogen activator (tPA), which promotes fibrinolysis
- The healthy endothelium also has a surface that discourages platelet adhesion.
- Endothelial cells synthesise and release von Willebrand factor (VWF), which is found in plasma and also within the subendothelium.
- Beneath the basement membrane lies the subendothelium, which is rich in:
- collagen
- elastin
- fibronectin
- tissue factor (TF)
- von Willebrand factor (VWF)
- Below this are the smooth muscle layer and the outer adventitia.
- If the endothelium is damaged, subendothelial collagen and VWF become exposed, allowing platelet adhesion and activation, which initiates thrombus formation.
- VWF is particularly important because it:
- binds platelets via the GPIb-IX-V receptor complex
- helps platelets adhere to the damaged vessel wall
- carries and stabilises factor VIII in the circulation
- The underlying smooth muscle can vasoconstrict after injury, which helps reduce blood loss.
🧫 Platelets
- Platelets are produced from megakaryocytes in the bone marrow.
- The normal platelet count is approximately 150–450 × 10⁹/L.
- The platelet lifespan is about 7–10 days.
- Platelets have a phospholipid membrane containing important glycoprotein receptors, which are essential for adhesion and aggregation.
- Platelet adhesion: when the endothelium is breached, platelets come into contact with subendothelial tissue. VWF binds to exposed collagen and to the platelet GPIb receptor complex, allowing platelets to adhere to the damaged vessel wall.
- Platelet activation: once adherent, platelets change shape and release granule contents such as:
- ADP
- serotonin
- calcium
- thromboxane A₂ (generated during activation)
- Platelet aggregation: activated platelets bind to each other via the GPIIb/IIIa receptor, using fibrinogen as a bridging molecule.
- Normal platelet number, intact platelet receptors, plasma fibrinogen, and calcium ions are all necessary for effective aggregation.
- This process forms the primary platelet plug, which is then reinforced by fibrin during coagulation.
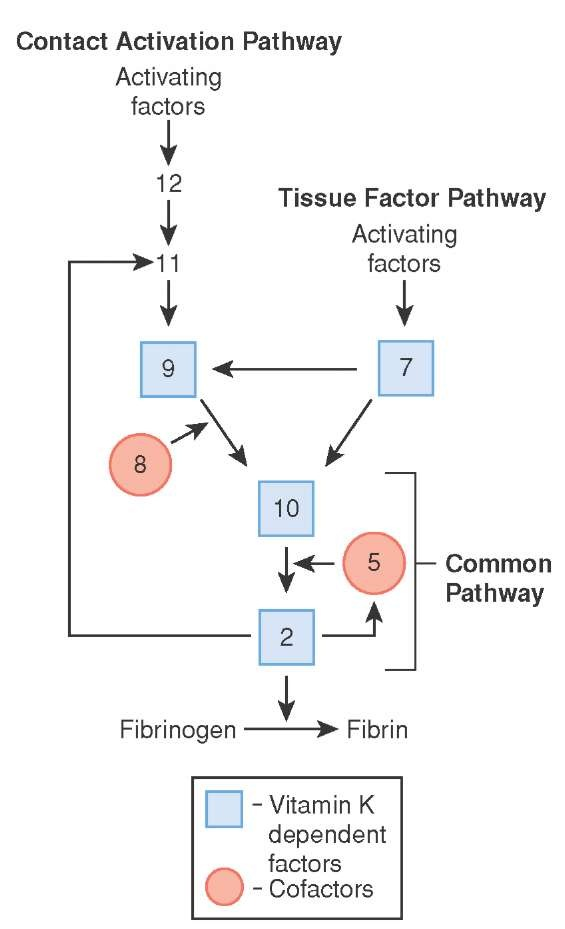
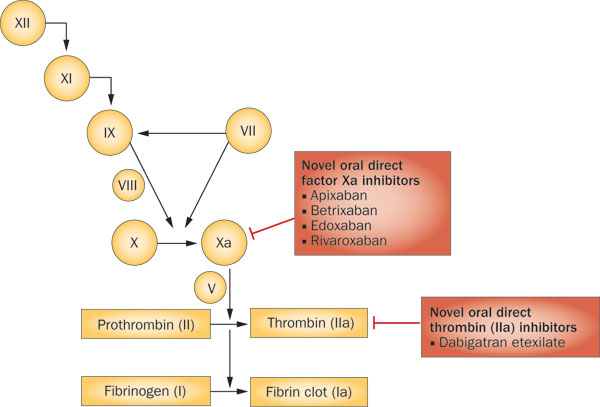
🧬 The Coagulation Phase
- In this phase, liquid blood is converted into a more stable, jelly-like clot.
- Coagulation occurs through a cascade of enzymatic reactions, in which each activated clotting factor activates the next step in the sequence.
- This allows the response to amplify rapidly, producing large amounts of thrombin.
- Thrombin is central to coagulation because it:
- converts fibrinogen into fibrin
- activates platelets
- amplifies other clotting factors
- Fibrin strands then stabilise the platelet plug to form a secondary haemostatic clot.
- Many clotting factors, especially factors II, VII, IX, and X, are vitamin K dependent.
- These factors are synthesised in the liver in precursor form and undergo gamma-carboxylation of glutamate residues.
- This vitamin K–dependent process allows them to bind calcium, which then helps anchor them to phospholipid surfaces, especially activated platelet membranes.
- This localisation is crucial because coagulation reactions occur most efficiently on phospholipid surfaces at the site of injury.
🧪 Laboratory Investigations
- Prothrombin Time (PT): assesses the extrinsic and common pathways, especially factors VII, X, V, II, and fibrinogen.
- Activated Partial Thromboplastin Time (APTT): assesses the intrinsic and common pathways, especially factors XII, XI, IX, VIII, X, V, II, and fibrinogen.
- Thrombin Time (TT): measures the conversion of fibrinogen to fibrin after addition of thrombin, and is prolonged in low fibrinogen states, dysfibrinogenaemia, or heparin exposure.
- Bleeding time: historically used to assess platelet–vessel wall interaction, but it is a poor predictor of surgical bleeding and is rarely used in modern practice.
🩹 Patterns of Bleeding
- Vascular or platelet disorders classically cause:
- petechiae
- purpura
- easy bruising
- epistaxis
- gum bleeding
- menorrhagia
- mucosal bleeding
- Coagulation factor deficiencies classically cause:
- deep tissue bleeding
- haematoma formation
- haemarthrosis
- post-operative or delayed bleeding
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
