| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Anatomy and Physiology of the Liver
Related Subjects: |AP of the Oesophagus |AP of the Diaphragm |AP of the Large Bowel (Colon, Rectum, Anal Canal) |AP of Small Bowel |AP of the Biliary system |AP of the Bone Marrow |AP of the Eye |AP of the Pharynx |AP of the Larynx |AP of the Ear |AP of the Nose |AP of Male Genitalia |AP of the Breast |AP of the Stomach |AP of the Rectum |AP of the Spleen |AP of the Liver
🫀 AP of the Liver (with Drug Metabolism + Paracetamol)
The liver is the body’s central metabolic and detoxification hub. Clinically, many problems map to: hepatocellular injury (ALT/AST), cholestasis (ALP/GGT), or synthetic/metabolic failure (INR, albumin, glucose, ammonia), often alongside portal hypertension as fibrosis advances.
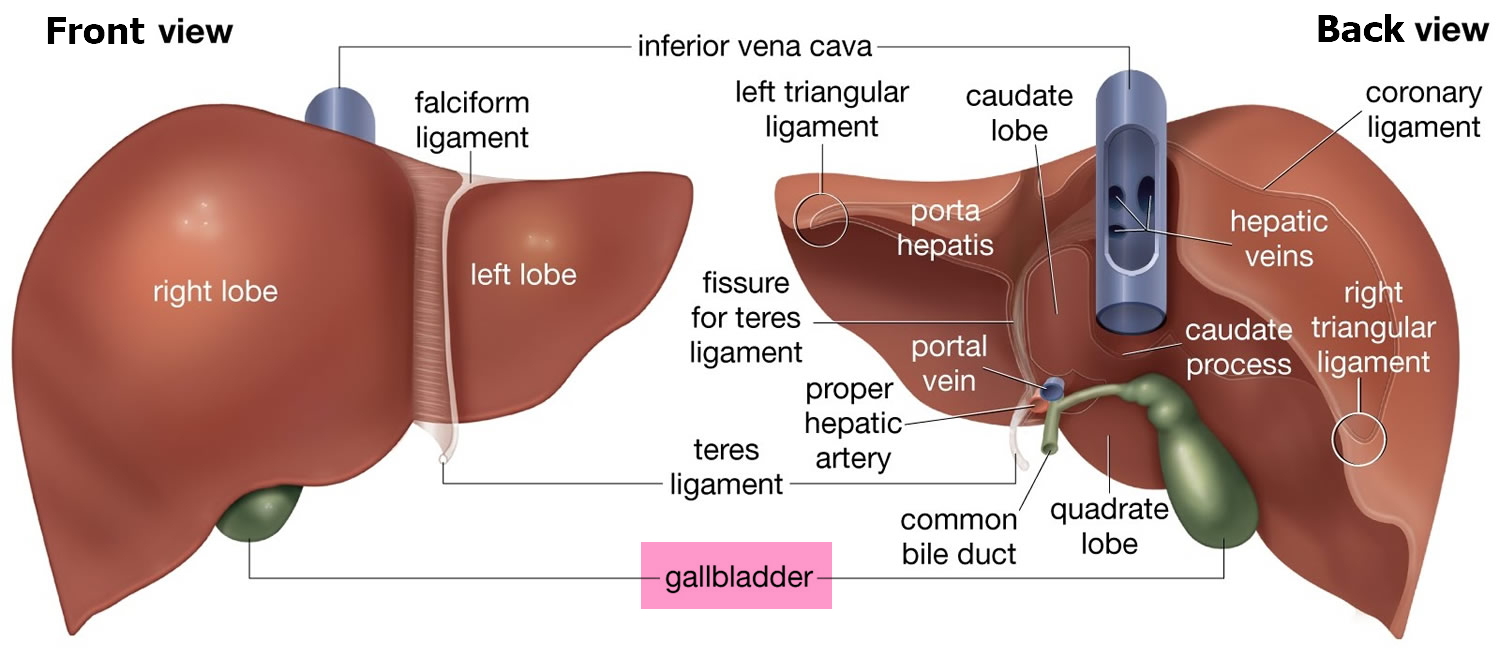
🧬 Introduction
- The liver weighs ~1.2–1.5 kg and performs hundreds of essential functions: metabolism, synthesis, storage, bile production, and detoxification.
- It lies mainly in the right upper quadrant beneath the diaphragm; pain is typically from capsular stretch (Glisson’s capsule), not hepatocytes themselves.
- Segmental anatomy (Couinaud segments) allows targeted resection and transplantation planning.
📍 Gross Anatomy and Couinaud Segments
Surface anatomy describes right and left lobes (falciform ligament). Functionally, the liver has 8 Couinaud segments, each with its own portal inflow, venous outflow, and biliary drainage.
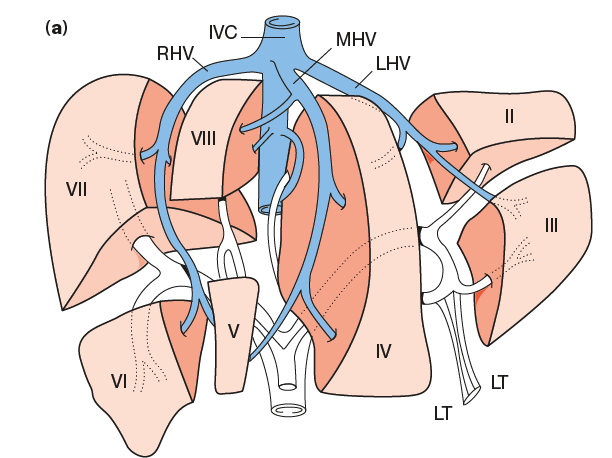
- I: Caudate (independent inflow; drains directly to IVC; can hypertrophy in cirrhosis).
- II–III: Left lateral (superior/inferior).
- IVa–IVb: Left medial (superior/inferior).
- V–VIII: Right lobe (anterior/posterior; inferior/superior).
🔬 Microscopic Anatomy (functional microcirculation)
- Lobule: Hepatocyte plates draining to a central vein.
- Portal triad: Portal venule + hepatic arteriole + bile ductule at the periphery.
- Sinusoids: Fenestrated vessels enabling rapid exchange (nutrients, drugs, toxins).
- Kupffer cells: Macrophages filtering portal blood (bacterial/endotoxin clearance).
- Space of Disse: Exchange interface; lymph formation site.
- Stellate cells: Store vitamin A; activation → collagen deposition → fibrosis/cirrhosis.
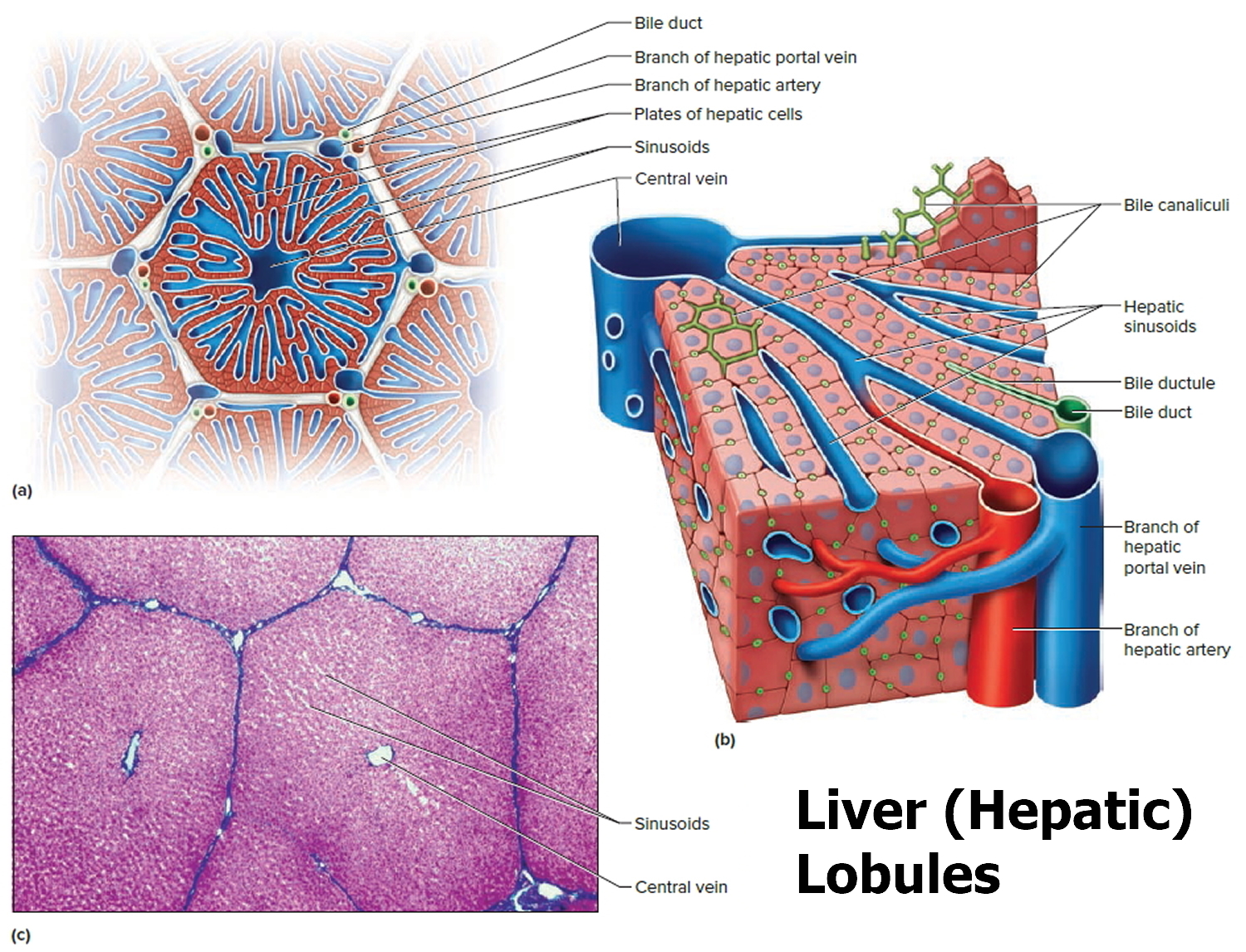
Fibrosis begins when stellate cells activate → collagen in the Space of Disse → sinusoidal “capillarisation” → reduced exchange + increased portal resistance (portal hypertension).
💉 Blood Supply and Portal Flow
- Portal vein (~75% flow): nutrient-rich blood from gut/spleen; delivers absorbed drugs/toxins first (first-pass effect).
- Hepatic artery (~25% flow): oxygen-rich blood from coeliac axis.
- Outflow: hepatic veins → IVC.
🟡 Bile Production and Flow
- Bile flows from hepatocytes → canaliculi → ducts → hepatic ducts → common hepatic duct → CBD → duodenum.
- Bile acids emulsify fats and enable absorption of fat-soluble vitamins (A, D, E, K); conjugated bilirubin is excreted via bile.
🗺️ Acinar Zonation (why injury patterns differ)
- Zone 1 (periportal): highest oxygen and nutrients; first exposed to portal blood (including some toxins).
- Zone 2: intermediate.
- Zone 3 (centrilobular): lowest oxygen but highest CYP450 activity → most vulnerable to hypoxic injury and reactive drug metabolites (classically paracetamol).
Zone 3 sits “furthest” from oxygenated inflow and is CYP450-rich - perfect setup for ischaemic hepatitis (shock liver) and toxic-metabolite injury.
⚙️ Liver Physiology - Expanded and Clinically Useful
🍞 Carbohydrate Handling
- Fed state: insulin-driven glycogenesis (glucose → glycogen).
- Fasting: glucagon/adrenaline-driven glycogenolysis (glycogen → glucose).
- Prolonged fasting/illness: gluconeogenesis from lactate (Cori cycle), glycerol, and amino acids.
- Clinical: severe liver failure → impaired gluconeogenesis → hypoglycaemia, especially in sepsis/alcohol misuse.
🧈 Lipid Handling
- Synthesises cholesterol (precursor for steroid hormones + bile acids).
- Packages triglycerides into VLDL for export; handles HDL pathways.
- β-oxidation produces energy; ketogenesis supports fasting physiology.
- Clinical: insulin resistance → hepatic triglyceride accumulation → MASLD/NAFLD.
🥚 Protein and Nitrogen Handling
- Synthesises albumin (oncotic pressure + drug binding) and many transport proteins.
- Synthesises clotting factors (II, VII, IX, X) and regulators (protein C/S) → explains raised INR in liver failure.
- Urea cycle converts ammonia to urea (prevents neurotoxicity).
- Clinical: impaired ammonia clearance → astrocyte swelling + neurotransmitter imbalance → hepatic encephalopathy.
🛡️ Immunology and Inflammation
- Kupffer cells filter portal blood, clearing bacteria/endotoxin (key barrier to gut translocation).
- Liver produces acute phase proteins (e.g. CRP) and complement components.
- Clinical: cirrhosis = immune dysfunction (susceptibility to infections like SBP).
💊 Drug Handling and Hepatic Biotransformation (very high-yield)
Most drugs are either metabolised in the liver, excreted in bile, or both. Hepatic handling depends on: hepatic blood flow, protein binding, intrinsic metabolic capacity, and biliary transport. Cirrhosis reduces functional hepatocyte mass and distorts microcirculation, so drug effects become unpredictable.
🏭 First-pass metabolism (why oral doses behave differently)
- Drugs absorbed from the gut enter the liver via the portal vein before reaching systemic circulation.
- High first-pass drugs can have low oral bioavailability; liver disease may increase bioavailability → higher plasma levels at “usual” doses.
- Examples (conceptual): some opioids, beta-blockers, nitrates, and calcium channel blockers have significant first-pass effects (drug-specific).
🧪 Phase I and Phase II metabolism
- Phase I (CYP450): oxidation/reduction/hydrolysis → may create reactive intermediates (important in toxicity).
- Phase II (conjugation): glucuronidation, sulphation, acetylation, methylation, glutathione conjugation → makes compounds water-soluble and safer for excretion.
- Cholestasis can impair biliary excretion → drug accumulation (especially with biliary elimination).
🔁 What changes in liver disease (practical prescribing physiology)
- ↓ Albumin → ↑ free fraction of highly protein-bound drugs (more effect/toxicity at same total level).
- Portosystemic shunting reduces first-pass clearance → oral doses behave “stronger”.
- ↓ CYP activity (variable) + ↓ hepatocyte mass → slower clearance for many drugs.
- Cholestasis → reduced biliary excretion.
- Ascites/edema → increased volume of distribution for hydrophilic drugs (complex dosing effects).
✅ Rule of thumb: in decompensated cirrhosis, start low, go slow, reassess frequently, and avoid hepatotoxic medicines where possible. Always consider renal function too (hepatorenal physiology).
🚑 Paracetamol (Acetaminophen) - Physiology of Toxicity
Paracetamol is usually safe at therapeutic doses because most is metabolised by glucuronidation and sulphation (Phase II pathways). A small fraction is metabolised via CYP450 (especially CYP2E1) to a reactive metabolite called NAPQI. NAPQI is normally detoxified by glutathione.
- Normal dose: conjugation pathways dominate; minimal NAPQI; glutathione easily neutralises it.
- Overdose: conjugation pathways saturate → more paracetamol diverted to CYP450 → excessive NAPQI → glutathione depletion → NAPQI binds hepatocyte proteins → oxidative injury and cell death.
- Where? Injury is typically centrilobular (Zone 3) due to high CYP activity and low oxygen reserve.
⚠️ Who is at higher risk of toxicity at lower doses?
- Chronic alcohol use (induces CYP2E1; may reduce glutathione stores).
- Malnutrition / fasting (reduced glutathione precursor availability).
- Enzyme-inducing drugs (some antiepileptics; depends on agent).
- Chronic liver disease (risk depends on reserve and nutrition; dosing must be cautious and individualised).
🧯 Why NAC works (mechanism, not just “because guidelines say so”)
- N-acetylcysteine (NAC) replenishes glutathione stores and provides sulphydryl groups to detoxify NAPQI.
- It also improves microcirculatory blood flow and acts as an antioxidant, which helps even when presentation is delayed.
Paracetamol toxicity is a classic example of liver physiology: Phase II saturation + Zone 3 CYP activity + glutathione depletion → predictable injury pattern. This is why timing, risk factors, and early antidote matter.
🩺 Clinical Relevance (how physiology shows up)
- Shock liver (ischaemic hepatitis): profound hypotension/hypoxia → marked ALT/AST rise; Zone 3 vulnerability.
- Cholestasis: pruritus + ALP/GGT rise; drug accumulation if biliary elimination impaired.
- Hepatic encephalopathy: ammonia and neurotoxin handling failure; precipitated by infection, GI bleed, constipation, sedatives.
- Cirrhosis: synthetic failure (INR, albumin), portal hypertension (varices/ascites), immune dysfunction.
✅ Makindo Exam Pearls
- Zone 3 is most vulnerable to hypoxia and paracetamol toxicity.
- INR is an early marker of reduced hepatic synthesis; albumin reflects chronic reserve.
- Paracetamol overdose: conjugation saturates → ↑ NAPQI → glutathione depletion → centrilobular necrosis; NAC restores glutathione.
- First-pass metabolism and reduced albumin explain why “standard doses” can be too strong in decompensated cirrhosis.
📌 Conclusion
The liver’s structure (segments, sinusoids, zonation) is inseparable from its function (metabolism, synthesis, bile, detox). Drug handling depends on blood flow, CYP activity, conjugation capacity, and biliary transport - all of which change in liver disease. Paracetamol toxicity neatly demonstrates these principles: Phase II saturation plus Zone 3 CYP metabolism creates NAPQI, and glutathione depletion drives predictable hepatocyte injury.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
