| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Barrett's oesophagus ✅
Related Subjects: |Achalasia |Oesophageal Carcinoma |Diffuse Oesophageal spasm |Diffuse Oesophageal Perforation - Rupture |Gastro-Oesophageal Reflux |Barrett's oesophagus
🧪 Barrett’s oesophagus is a premalignant condition in which normal distal oesophageal squamous epithelium is replaced by specialised intestinal metaplasia (columnar epithelium with goblet cells) due to chronic reflux. ⚠️ It increases the risk of oesophageal adenocarcinoma, particularly in the presence of dysplasia. 🧠 Key concept: metaplasia → dysplasia → carcinoma sequence.
📌 Definition (BSG-aligned)
- Endoscopically visible columnar-lined oesophagus ≥1 cm above the GOJ.
- Requires histological confirmation of intestinal metaplasia (goblet cells).
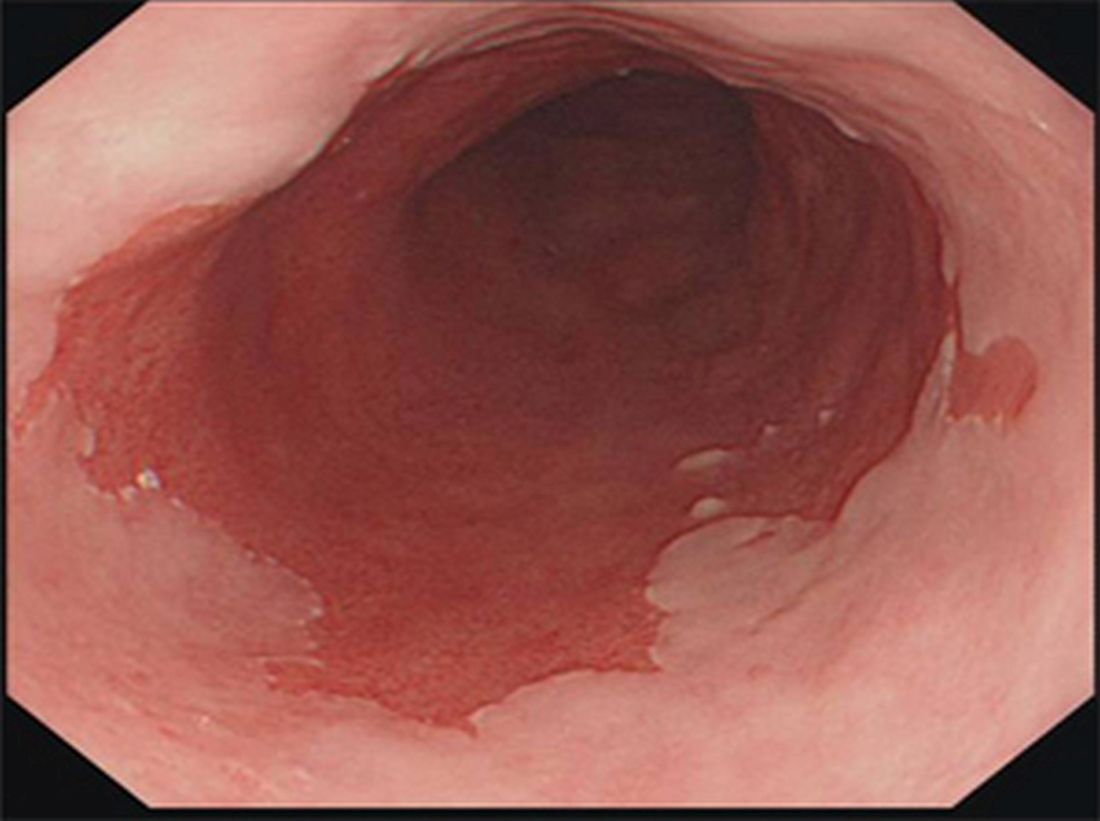
- 🍣 Appears as salmon-pink mucosa replacing pale squamous lining.
🧬 Pathophysiology
- 🔥 Chronic acid ± bile reflux → epithelial injury.
- 🔄 Healing via metaplasia (columnar epithelium more acid-resistant).
- ⚠️ Genetic instability → progression:
- Metaplasia → low-grade dysplasia → high-grade dysplasia → adenocarcinoma
⚠️ Risk Factors
- 👨 Male sex
- 🎂 Age >50
- ⚖️ Central obesity (↑ intra-abdominal pressure → reflux)
- 🚬 Smoking
- 🔥 Chronic GORD
- 👨👩👧 Family history of oesophageal adenocarcinoma
🔥 Risk of Malignant Transformation
| Finding | Relative Risk |
|---|---|
| Non-dysplastic Barrett’s | Low (~0.1–0.3%/year) |
| Low-grade dysplasia | Moderate (~0.5–1%/year) |
| High-grade dysplasia | High (significant short-term risk) |
| Long-segment (>3 cm) | ↑ risk |
🔎 Investigations
- 🔬 Upper GI endoscopy (OGD):
- Salmon-coloured mucosa extending proximally from GOJ
- Document Prague classification (extent: C & M length)
- 🧫 Biopsy (Seattle protocol):
- Quadrantic biopsies every 2 cm
- Every 1 cm if dysplasia suspected
- 🧬 Histology:
- Confirm goblet cells (intestinal metaplasia)
- Grade dysplasia
- 🎨 Adjuncts: Chromoendoscopy / narrow-band imaging (target dysplasia)
💊 Management (NICE + BSG aligned)
- 🍽️ Lifestyle: Weight loss, smoking cessation, reduce alcohol, elevate head of bed
- 💊 Acid suppression:
- PPI long-term (symptom control + may reduce progression risk)
- 🔍 Endoscopic surveillance: (BSG 2014 / NICE CG106 principles)
- <3 cm (short segment): every 3–5 years
- ≥3 cm (long segment): every 2–3 years
- 🔥 Dysplasia management:
- Low-grade dysplasia: Confirm by expert pathologist → RFA preferred
- High-grade dysplasia: Endoscopic eradication therapy (EMR + RFA)
- 🔪 Surgery (oesophagectomy):
- Reserved for selected cases (e.g. invasive cancer, unsuitable for endoscopic therapy)
📏 NICE/BSG Surveillance Nuances (Exam High-Yield)
- 🧠 Surveillance only if intestinal metaplasia confirmed
- ⚠️ Dysplasia diagnosis should be confirmed by 2 expert GI pathologists
- 🔁 “Indefinite for dysplasia” → optimise PPI → repeat endoscopy (~6 months)
- 🚫 No population screening in UK (only targeted case-finding)
📚 References
- BSG Guidelines – Barrett’s Oesophagus
- NICE CG106 – Barrett’s Oesophagus & Oesophageal Cancer
- NICE NG1 – Dyspepsia and GORD
🧑⚕️ Teaching Commentary
Barrett’s oesophagus represents an adaptive response to chronic acid exposure, where squamous epithelium is replaced by columnar epithelium better suited to an acidic environment. The clinical importance lies in the dysplasia–carcinoma sequence, with risk stratified by histology rather than symptoms. This explains why surveillance is targeted at those with confirmed intestinal metaplasia and why dysplasia mandates escalation to endoscopic therapy. In UK practice, the shift has been toward endoscopic eradication (RFA ± EMR) rather than surgery for dysplasia, reflecting excellent outcomes with organ preservation. Always remember: the biggest modifiable driver is ongoing reflux, making PPI therapy and lifestyle optimisation foundational.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
