| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Parkinson disease
Related Subjects: Multiple System Atrophy (MSA) |Parkinson Plus syndromes |Parkinsonism |Idiopathic Parkinson disease |Acutely Ill Patient with Parkinson's disease |Progressive Supranuclear Palsy |Drug Induced Parkinson disease |Neuroleptic Malignant Syndrome
🧠 Parkinson's Disease (PD) is a chronic, progressive neurological disorder affecting movement and non-motor functions. 👴 Most often seen in older adults, though genetic forms can present earlier.
ℹ️ About
- 🌀 Idiopathic PD: Tremor, rigidity, bradykinesia.
- 👵 More common in elderly, but younger onset with genetic variants.
🧬 Aetiology
- ⬇️ Dopaminergic neurons lost in the substantia nigra.
- ⚡ Disrupted communication in the basal ganglia.
- 🧩 Lewy bodies (α-synuclein inclusions) hallmark of PD pathology.
Pathology: Braak Staging
- 📈 Pathology spreads stepwise: brainstem → midbrain → cortex.
- 🚽 Early = constipation, REM sleep disorder; 🤲 Later = tremor, rigidity.
Genetics
- 🧬 PARK1 (α-synuclein): Early-onset, autosomal dominant.
- 🧬 PARK2 (parkin): Autosomal recessive, good response to L-Dopa.
🩺 Progressive Clinical Features
- ✋ Tremor: “Pill-rolling” resting tremor.
- 🐢 Bradykinesia: Slowness, difficulty initiating movement.
- 🪵 Rigidity: Limb stiffness, cogwheeling.
- ⚖️ Postural instability: Falls, poor balance.
- 🌙 Non-motor: depression, constipation, sleep disturbance, anosmia.
Stages of PD
- 1️⃣ Mild unilateral tremor.
- 2️⃣ Bilateral symptoms, slower movement.
- 3️⃣ Falls, significant motor difficulty.
- 4️⃣ Severe disability, daily assistance required.
- 5️⃣ Bedridden/wheelchair, hallucinations possible.
Diagnostic Imaging
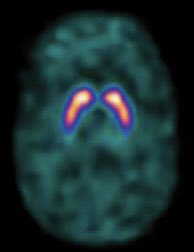
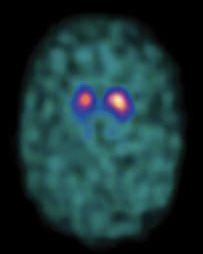
- 🧲 MRI: Exclude mimics (vascular PD, NPH).
- 📊 DaT Scan: Differentiates PD from essential tremor.
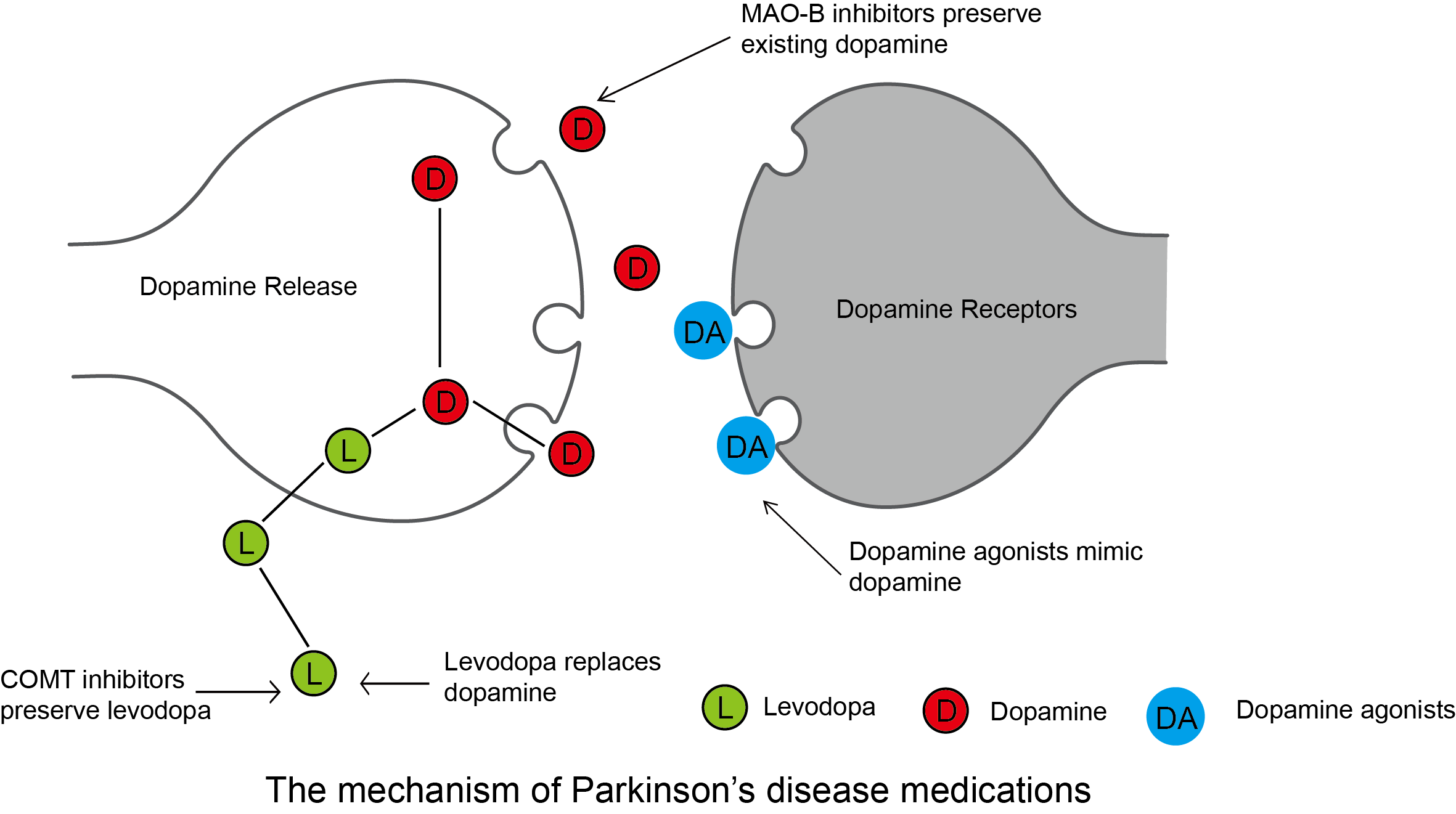
Pharmacological Management
- 💊 Levodopa + Carbidopa (Sinemet): Gold standard, best for motor symptoms.
- 🟤 COMT inhibitors: Prolong levodopa action (e.g. entacapone).
- 🟢 Dopamine agonists: Ropinirole, pramipexole (younger patients).
- 🔵 MAO-B inhibitors: Rasagiline, selegiline (mild disease).
- 🟣 Anticholinergics: Tremor-dominant PD (younger patients).
- 🟡 Amantadine: Useful for dyskinesias.
🧠 About Parkinson’s Drug Management
- Parkinson’s disease results from dopamine deficiency in the basal ganglia, leading to tremor, rigidity, bradykinesia, and postural instability.
- The main goal of therapy is to restore dopaminergic tone, reduce motor and non-motor symptoms, and improve quality of life.
- Treatment strategies are individualised based on patient age, disease stage, cognitive function, frailty, and comorbidities.
- Combination therapy is often required as the disease progresses, and management should be supervised by a specialist in movement disorders.
💊 Main Drug Classes & Dosing
- 1️⃣ Levodopa + DDC inhibitor (co-careldopa, co-beneldopa)
- Mechanism: Levodopa is converted to dopamine in the brain; carbidopa or benserazide blocks peripheral metabolism.
- First-line in patients >70 years or with cognitive impairment.
- Dose:
- Start low (e.g. co-careldopa 12.5/50 mg TDS) and titrate gradually.
- Typical maintenance: 50/200 mg TDS–QDS, adjusted to clinical response.
- Modified formulations:
- Rytary (IPX203): extended-release capsule for smoother “on” time.
- LCIG (Duopa/Duodopa): intestinal gel via pump for continuous delivery.
- Inhaled Levodopa (Inbrija): rescue option for sudden “off” episodes, onset within 10 minutes.
- ND0612: subcutaneous infusion for 24-hour steady levels (emerging therapy).
- ⚠️ Common side effects: dyskinesia, orthostatic hypotension, hallucinations, nausea, “on–off” fluctuations with long-term use.
- 2️⃣ Dopamine Agonists (pramipexole, ropinirole, rotigotine patch, apomorphine)
- Mechanism: Directly stimulate dopamine receptors (mainly D2/D3), bypassing presynaptic neurons.
- Use: Monotherapy in early PD for younger adults, or as add-on to levodopa in advanced disease.
- Examples & Doses:
- Pramipexole: 0.125 mg TDS → titrate to max 1.5 mg TDS.
- Ropinirole: 0.25 mg TDS → increase; XL forms available for once-daily dosing.
- Rotigotine patch: start 2 mg/24 h → titrate to max 16 mg/24 h.
- Apomorphine: subcutaneous rescue injection or continuous infusion (specialist use only).
- ⚠️ Side effects: impulse control disorders (gambling, hypersexuality), hallucinations, nausea, ankle oedema, sudden sleep attacks.
- Note: Start low, titrate slowly, monitor mood and sleep; avoid abrupt withdrawal to prevent dopamine agonist withdrawal syndrome.
- 3️⃣ MAO-B Inhibitors (selegiline, rasagiline, safinamide)
- Mechanism: Inhibit monoamine oxidase-B, decreasing dopamine breakdown in the brain.
- Use: Early PD (as monotherapy) or adjunct therapy to prolong levodopa effect and reduce “off” episodes.
- Dosing:
- Selegiline: 5 mg OD or BD.
- Rasagiline: 1 mg OD.
- Safinamide: 50 mg OD → may increase to 100 mg OD.
- ⚠️ Side effects: insomnia, headache, mild hallucinations; rare hypertensive crisis with tyramine-rich foods.
- Note: Avoid combining with serotonergic antidepressants or opioids due to serotonin syndrome risk.
- 4️⃣ COMT Inhibitors (entacapone, opicapone, tolcapone)
- Mechanism: Inhibit catechol-O-methyltransferase (COMT), prolonging levodopa’s half-life and “on” time.
- Use: Always as adjunct to levodopa when wearing-off occurs.
- Dosing:
- Entacapone: 200 mg with each levodopa dose (max 10/day).
- Opicapone: 50 mg once nightly (newer, long-acting).
- Tolcapone: 100–200 mg TDS; effective but limited by hepatotoxicity.
- ⚠️ Side effects: diarrhoea, urine discolouration (orange), dyskinesia, hepatotoxicity (tolcapone-monitor LFTs).
- 5️⃣ Amantadine
- Mechanism: NMDA receptor antagonist; enhances dopamine release and reduces reuptake.
- Dose: 100 mg OD–BD (max 300 mg/day); extended-release (Gocovri ER) once daily at bedtime.
- Use: Helps control levodopa-induced dyskinesias and provides mild symptomatic benefit.
- ⚠️ Side effects: hallucinations, livedo reticularis (mottled skin), ankle oedema, confusion, blurred vision.
- Note: Dose reduction required in renal impairment and elderly patients.
- 6️⃣ Anticholinergics (trihexyphenidyl, procyclidine)
- Mechanism: Block muscarinic receptors, restoring balance between dopamine and acetylcholine.
- Dose: Trihexyphenidyl 1 mg OD–BD (max 15 mg/day); adjust slowly.
- Use: Tremor-predominant PD in young, cognitively intact patients.
- ⚠️ Side effects: confusion, memory impairment, urinary retention, constipation, dry mouth - avoid in elderly.
- 7️⃣ Adjunct & Emerging Agents
- Adenosine A2A antagonist (Istradefylline): Adjunct to levodopa in patients with “off” episodes; reduces "off" time and improves mobility.
- GLP-1 receptor agonists (Exenatide, Lixisenatide): Under investigation for neuroprotective and disease-modifying benefits.
- Gene therapy (AADC vector, AXO-Lenti-PD): Experimental approaches to restore dopamine synthesis.
🧩 Key Points
- Levodopa remains the gold standard for symptomatic control.
- Long-acting and continuous delivery systems (LCIG, ND0612, Rotigotine patch) help minimize “off” time and fluctuation.
- Adjunct agents (MAO-B, COMT inhibitors, adenosine antagonists) prolong levodopa benefit.
- Tailor therapy to symptom pattern, lifestyle, and patient goals - avoid polypharmacy where possible.
- Monitor regularly for side effects such as dyskinesia, hallucinations, orthostatic hypotension, and impulse control disorders.
⚙️ Advanced / Rescue Therapies
- LCIG (Duopa/Duodopa): Continuous intrajejunal infusion for advanced PD with severe motor fluctuations.
- Apomorphine Pump: Delivers continuous dopamine agonist stimulation; used for complex “off” episodes.
- Inhaled Levodopa (Inbrija): Rapid rescue during sudden wearing-off; onset within 10 minutes.
- Deep Brain Stimulation (DBS): Non-pharmacologic option for selected patients unresponsive to medications.
✅ Summary
Parkinson’s disease management requires a stepwise, individualized approach. Start with levodopa-based therapy in older or cognitively impaired patients, and dopamine agonists or MAO-B inhibitors in younger individuals. Add COMT inhibitors or amantadine as the disease progresses to reduce fluctuations and dyskinesia. Advanced and long-acting formulations-both oral and infusion-based-are now enabling more stable motor control and improved quality of life. Levodopa remains the most effective treatment for Parkinson’s disease. Long-acting and continuous infusion forms improve symptom stability, while adjuncts-such as dopamine agonists, MAO-B inhibitors, and COMT inhibitors-help control motor fluctuations. Therapy should always be individualized to minimize side effects and optimize quality of life.
📋 NICE Simplified Pathway
- 👴 Older (>70) or cognitive impairment → Levodopa.
- 🧑 Younger (<70), no cognitive issues → Dopamine agonist or MAO-B inhibitor.
- ⚡ Escalate stepwise with COMT inhibitors, dopamine agonists, or amantadine if fluctuations develop.
- Specialist referral at all treatment stages.
🚑 Management When Unable to Swallow (NBM)
- 💧 Dispersible levodopa (Madopar® dispersible): can be given via NG tube.
- 🩹 Rotigotine patch: provides continuous dopaminergic stimulation.
- 💉 Apomorphine (SC injection/infusion): specialist initiation, useful for acute rescue.
- 📡 Duodopa® (levodopa–carbidopa intestinal gel): continuous PEG-J infusion.
- ⚠️ Never stop dopaminergic drugs suddenly → risk of Parkinsonism–Hyperpyrexia Syndrome.
⚠️ Key Cautions & Monitoring
- Impulse control disorders (gambling, hypersexuality, binge eating) with dopamine agonists.
- Hallucinations & psychosis with dopaminergic drugs.
- Postural hypotension with levodopa and dopamine agonists.
- Monitor LFTs with tolcapone.
- Adjust doses in renal impairment for amantadine.
Surgical
- 🧩 Deep Brain Stimulation (DBS): For advanced disease, subthalamic nucleus/globus pallidus targets.
Multidisciplinary Care
- 👩⚕️ Nurse specialist: medication timing, education.
- 🏃 Physiotherapy: balance, gait training.
- 🗣️ SLT: dysarthria, dysphagia.
- 🛠️ OT: adaptive aids, safety at home.
Hospital/Acute Considerations
- ⏰ Never delay PD meds (risk of severe deterioration, even NBM use NG or rotigotine patch).
- 🚫 Avoid antidopaminergic drugs (metoclopramide, haloperidol, prochlorperazine).
Non-Motor Complications
- 🚽 Constipation → macrogol, fluids, mobility.
- 📉 Orthostatic hypotension → fludrocortisone, midodrine.
- 🦠 Recurrent UTIs → hydration, constipation prevention.
- 👻 Psychosis/hallucinations → quetiapine, clozapine.
- 🧩 Dementia → rivastigmine (watch for tremor worsening).
Advance Care Planning
- 📝 Advance directives, lasting power of attorney.
- 🤝 Signposting to community support and respite care.
References
Cases - Parkinson’s Disease with Complications
- Case 1 - Motor Fluctuations (“Wearing Off”) ⏳: A 68-year-old man on levodopa for 7 years reports that tremor and rigidity return 3 hours after each dose. He experiences “on–off” fluctuations through the day. Complication: Motor fluctuations due to long-term levodopa use. Management: Shorten levodopa dosing intervals; add COMT inhibitor (entacapone) or dopamine agonist.
- Case 2 - Dyskinesias 💃: A 62-year-old woman develops involuntary writhing movements of her trunk and arms about 30 minutes after each levodopa dose. Complication: Levodopa-induced dyskinesia. Management: Lower levodopa dose; add amantadine; consider deep brain stimulation if severe.
- Case 3 - Parkinson’s Disease Dementia 🧠: A 75-year-old man with 10 years of PD develops progressive memory loss, visual hallucinations, and difficulty with daily tasks. Complication: Parkinson’s disease dementia. Management: Rivastigmine (first-line cholinesterase inhibitor); review dopaminergic drugs that worsen hallucinations.
- Case 4 - Autonomic Dysfunction 💧: A 70-year-old woman with PD reports frequent faints on standing, constipation, and urinary urgency. BP falls from 135/80 supine to 95/60 standing. Complication: Autonomic dysfunction (orthostatic hypotension, bladder involvement). Management: Non-pharmacological (slow position changes, compression stockings, ↑ salt/fluid); consider fludrocortisone or midodrine.
- Case 5 - Impulse Control Disorder 🎰: A 60-year-old man on pramipexole for PD develops compulsive gambling and hypersexuality, which his wife finds distressing. Complication: Dopamine agonist–induced impulse control disorder. Management: Reduce/stop dopamine agonist; switch to levodopa-based regimen; behavioural support.
Teaching Commentary 🧠
Parkinson’s disease complications can be divided into: - Motor: fluctuations (“wearing off”), dyskinesias. - Neuropsychiatric: dementia, hallucinations, depression, impulse control disorders. - Autonomic: postural hypotension, constipation, bladder dysfunction, erectile dysfunction. - Sleep: REM sleep behaviour disorder, insomnia. Complications often reflect both disease progression and treatment side effects. Management requires balancing dopaminergic therapy, adding adjuncts (COMT/MAO-B inhibitors), and addressing non-motor symptoms with MDT support. Deep brain stimulation is an option in selected patients with refractory motor fluctuations.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
