| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Brain Tumours ✅
Related Subjects: |Brain tumours |Astrocytomas |Brain Metastases |Tuberous sclerosis |Turcot's syndrome |Lhermitte Duclos Disease |Oligodendroglioma |Acute Hydrocephalus |Intracranial Hypertension |Primary CNS Lymphoma (PCNSL) |Astrocytomas |Glioblastoma
🧠 Brain tumours can be primary (arising in the brain) or secondary (metastatic from other cancers). Symptoms reflect both the local effects of the lesion and raised intracranial pressure (ICP). Diagnosis relies on neuroimaging and histological confirmation.
🔎 Overview
- Primary tumours arise from glial cells, neurons, or meningeal tissue.
- Secondary tumours commonly spread from lung, breast, kidney, thyroid, testes, or melanoma.
- Neuroimaging (MRI/CT) is essential; biopsy confirms histology.
🧬 Types of Brain Tumour
- Primary: Gliomas (astrocytomas, oligodendrogliomas, glioblastomas), meningiomas.
- Secondary (metastatic): From lung, breast, colorectal, kidney, testis, melanoma.
- HIV-related: Primary CNS lymphoma (B-cell type).
🧾 Clinical Presentation
- Headache – classically worse in the morning, aggravated by straining/coughing.
- Seizures, personality/behaviour change, stroke-like deficits, or coma.
- Raised ICP → nausea, papilloedema, visual disturbances.
📍 Localising Features
- Olfactory groove tumour: Anosmia, disinhibition, personality change.
- Cavernous sinus tumour: Ophthalmoplegia (CN III, IV, VI, V1, V2 involvement).
- Foster Kennedy syndrome: Ipsilateral optic atrophy + contralateral papilloedema (often olfactory groove meningioma).
- Pituitary adenoma: Hypopituitarism, headache, bitemporal hemianopia.
- Pineal region tumour: Parinaud’s syndrome (loss of upward gaze).
🧪 Investigations
- Bloods: FBC, U&E, LFT, ESR/CRP, ± HIV testing.
- CXR: To exclude primary lung cancer in suspected metastasis.
- Tumour markers: AFP, CEA, CA125, PSA, S-100 depending on suspicion.
- Imaging:
- CT head with contrast – tumour enhancement due to BBB breakdown.
- MRI with gadolinium – superior for staging and characterisation.
- Biopsy: Required for histology and treatment planning.
🖼️ Example Imaging
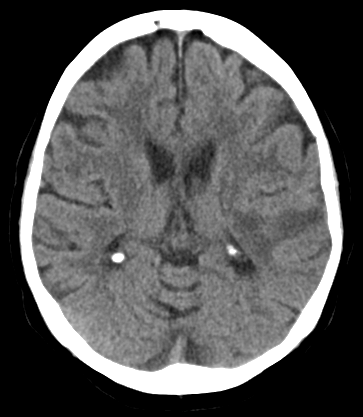
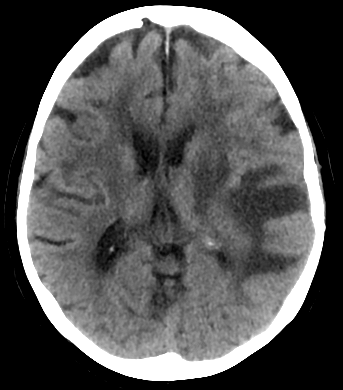
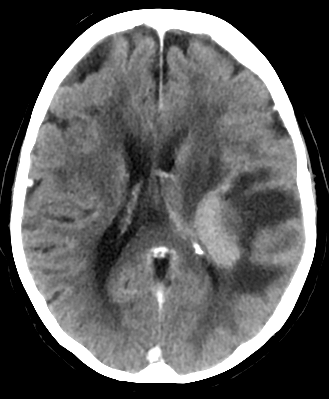
💊 Management
- Surgery: For histology, debulking, or relieving hydrocephalus.
- Chemotherapy: e.g., temozolomide for gliomas.
- Radiotherapy:
- External beam (e.g., medulloblastoma, CNS lymphoma).
- Stereotactic radiosurgery for small meningiomas.
- Supportive care:
- Anticonvulsants for seizure prophylaxis.
- Dexamethasone for cerebral oedema.
- Neuro-oncology MDT for holistic care.
- Palliative care in advanced/inoperable cases.
💊 Brain Tumour Medications & Supportive Therapy
| Drug / Therapy | Indication | Typical Dose | Common Side Effects | Key Contraindications / Cautions |
|---|---|---|---|---|
| Temozolomide (oral alkylating chemo) | High-grade gliomas (GBM, astrocytomas) | 75 mg/m²/day during radiotherapy; 150–200 mg/m²/day × 5 days/month post-radiotherapy | Nausea, vomiting, myelosuppression, fatigue, lymphopenia | Severe myelosuppression, pregnancy, caution in hepatic impairment |
| Carboplatin / Cisplatin | Pediatric brain tumours, medulloblastoma, CNS metastases | Weight / BSA-based dosing per protocol | Nephrotoxicity, ototoxicity, myelosuppression, nausea | Renal impairment, hearing loss, pregnancy |
| Dexamethasone (corticosteroid) | Peritumoral cerebral oedema; raised ICP | 4–16 mg/day orally or IV, titrated to symptoms | Hyperglycaemia, insomnia, weight gain, immunosuppression, GI upset | Uncontrolled diabetes, systemic infections, peptic ulcer disease |
| Anticonvulsants (Levetiracetam, Phenytoin) | Seizure prophylaxis / treatment in brain tumours | Levetiracetam 500–1500 mg BID; Phenytoin 100 mg TDS, adjust per levels | Drowsiness, dizziness, irritability, rash (esp. phenytoin) | Allergy, severe hepatic impairment (phenytoin), pregnancy (phenytoin caution) |
| Radiotherapy | Adjuvant therapy post-surgery; small meningiomas, medulloblastoma, CNS lymphoma | External beam: fractionated doses per tumour type; SRS: single fraction 12–24 Gy | Fatigue, alopecia, skin erythema, nausea; long-term: cognitive decline, radiation necrosis | Pregnancy, prior high-dose cranial irradiation |
| Supportive / Symptomatic Therapy | Edema, nausea, pain, mood disturbance | Varies: e.g., Mannitol 0.25–1 g/kg IV for acute raised ICP; antiemetics as needed | Fluid/electrolyte shifts, hypotension (mannitol), sedation (antiemetics/opioids) | Severe renal failure (mannitol), hypersensitivity to drugs |
💡 Notes: - Always use multidisciplinary discussion (neuro-oncology MDT) for individualised treatment. - Dexamethasone should be tapered as soon as oedema improves to minimise side effects. - Chemotherapy dosing varies with protocol, tumour type, and patient tolerance; monitor blood counts closely. - Anticonvulsants may interact with chemotherapeutic agents (esp. phenytoin/carbamazepine).
💡 Exam Pearls
• Suspect brain tumour if “stroke” symptoms evolve over days/weeks. • Red flags: morning headache, seizures, personality change, progressive neurological deficits. • MRI with contrast + biopsy = gold standard for diagnosis.
📚 References & NICE Guidance
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
