| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Hydrogen and other Bonds
Related Subjects: |Carbohydrates |Hydrogen and other Bonds
🌍 Life depends on chemistry: every heartbeat, thought, and breath reflects countless atoms of oxygen, carbon, hydrogen, and other elements forming and breaking bonds. ⚡ These dynamic interactions create the structure of biomolecules and drive metabolism, energy transfer, and signalling.
🔗 Types of Chemical Bonds
- ⚡ Ionic Bonds:
Ions are charged atoms formed when one atom donates or accepts electrons.
- Positive = cation (e.g., Na⁺)
- Negative = anion (e.g., Cl⁻)
- Opposite charges attract → form ionic bonds (e.g., NaCl crystals).
- Biological relevance: Key in nerve conduction, muscle contraction, and electrolyte balance.
- 🔗 Covalent Bonds:
Electrons are shared between atoms.
- Nonpolar covalent bonds: Equal sharing of electrons → electrically neutral molecules (e.g., O₂, CH₄). 🧪 Basis of lipid membranes (hydrophobic tails).
- Polar covalent bonds: Unequal sharing → “poles” of charge (e.g., H₂O). 💧 This underlies solubility and hydrogen bonding.
- Biological relevance: Stable backbone of DNA, proteins, carbohydrates.
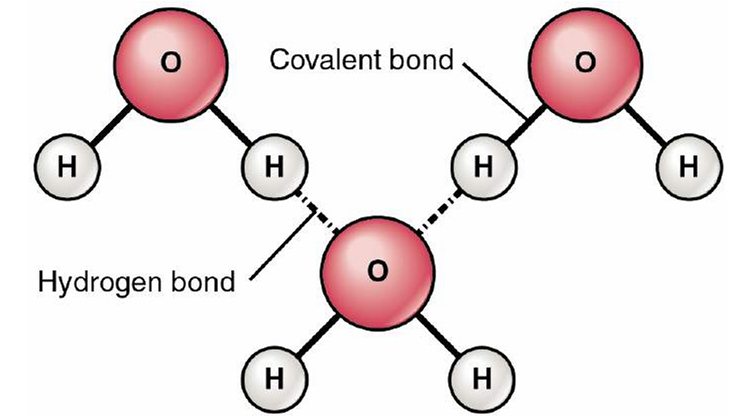
- 💧 Hydrogen Bonds:
A weak but vital interaction when a hydrogen atom covalently bound to an electronegative atom (O, N) is attracted to another electronegative atom.
- Weaker than covalent/ionic bonds but crucial for life.
- Stabilises DNA double helix, protein secondary structures (α-helix, β-sheet).
- Gives water its unique properties: high boiling point, surface tension, excellent solvent.
🧬 Integration in Biology
In living organisms, bonds constantly form and break to allow flexibility and energy flow:
- 💧 Water: Polar covalent bonds → hydrogen bonding → solvent of life.
- 🧂 Ionic interactions: Salt (NaCl) dissolves via ion-dipole interactions with water.
- 🥑 Nonpolar covalent bonds: Hydrophobic lipids cluster in membranes → essential for cell structure.
- 🧩 Proteins: Hydrogen bonds + ionic interactions stabilise folding and enzyme function.
📊 Clinical & Physiological Relevance
- ⚡ Electrolyte imbalance: Low Na⁺, K⁺, or Ca²⁺ disrupt ionic gradients → arrhythmias, seizures.
- 🧬 DNA damage: Breaking hydrogen bonds → mutation or apoptosis.
- 🌡️ Fever: Alters hydrogen bonding in proteins → denaturation at high temperatures.
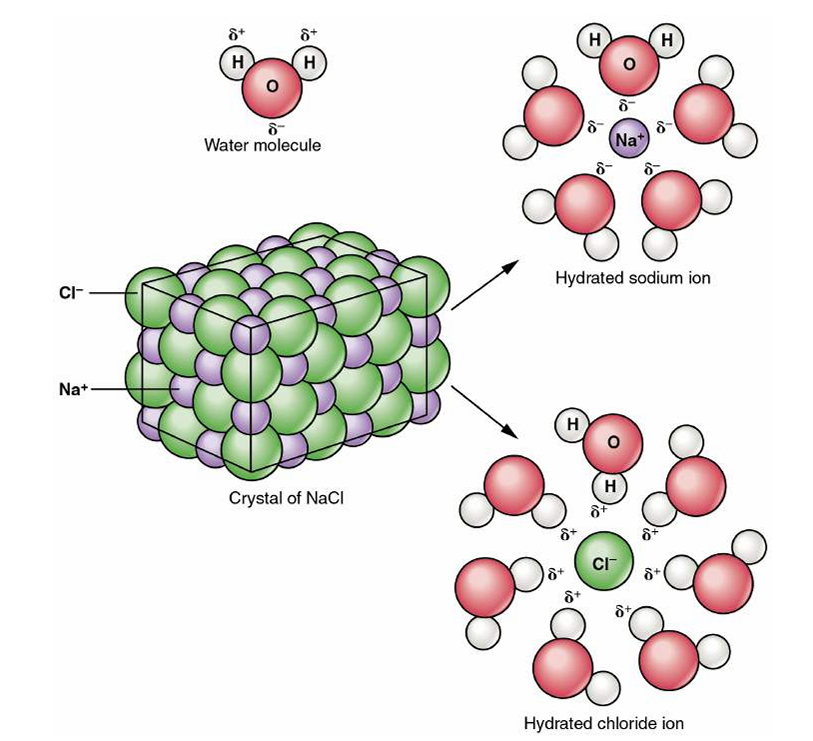
🧂 Dissociation of Sodium Chloride in Water
NaCl does not dissolve as intact molecules but dissociates into Na⁺ cations and Cl⁻ anions, each surrounded by water molecules (hydration shells) → stabilising ions in solution. 🔑 This principle explains conduction in nerves and osmotic gradients.
✅ Conclusion
From ionic Na⁺/Cl⁻ gradients powering nerve impulses, to hydrogen bonds shaping DNA and proteins, chemical bonds are the language of biology. 🔑 Understanding these interactions helps explain everything from cell signalling and drug action to genetic inheritance and disease.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
