| Download the amazing global Makindo app: ✅ Means NICE/National Guidelines 2026 compliant Android | Apple | |
|---|---|
| MEDICAL DISCLAIMER: Educational use only. Not for diagnosis or management. See below for full disclaimer. |
Anatomy and Physiology of the Heart
Related Subjects: | AP of the Heart | Cardiac Embryology | AP of the Coronary Arteries
🫀 The heart is a muscular, cone-shaped pump in the middle mediastinum, enclosed by the pericardium and resting on the diaphragm. It is oriented obliquely: the apex points downwards and left (classically at the 5th intercostal space, mid-clavicular line), while the base faces posteriorly toward the vertebral column and is formed mainly by the left atrium. Functionally, the heart acts as two pumps in series-right heart to the lungs (low pressure) and left heart to the body (high pressure)-with tight coupling between electrical activation, valve mechanics, and haemodynamics.
📌 Exam Tip: Apex beat normally felt at 5th intercostal space, MCL. Lateral/inferior displacement suggests LV dilatation or LVH (e.g., chronic hypertension, aortic stenosis), and a heaving character suggests pressure overload.
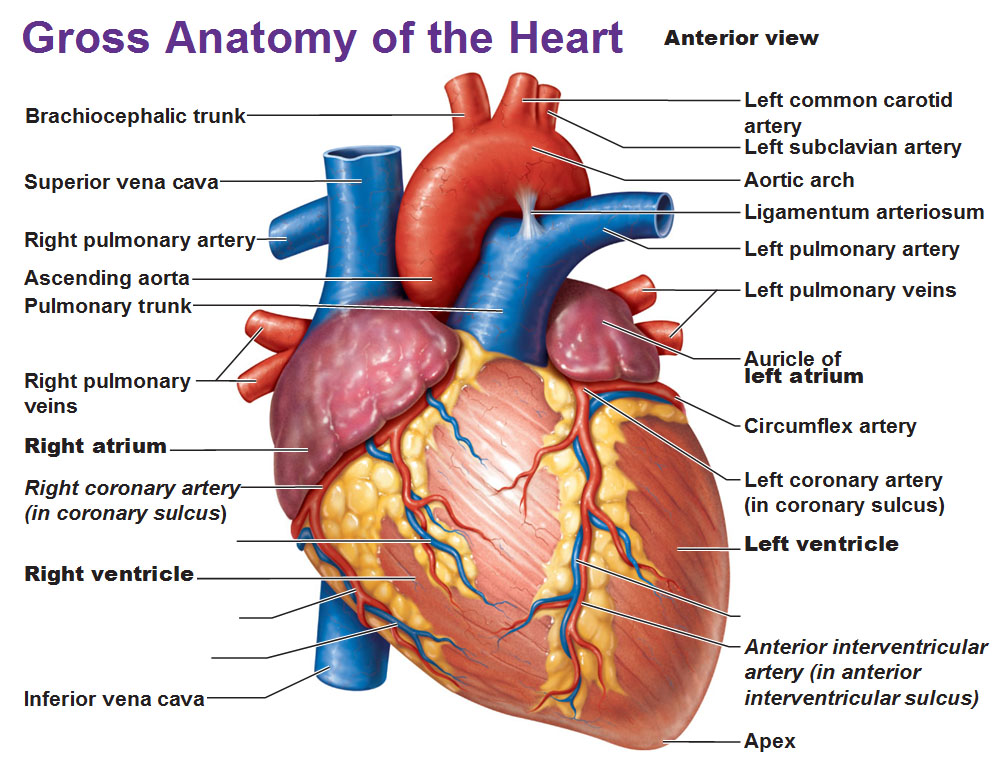
🏗️ Structural Overview
- Four chambers:
- 🫀 Right atrium (RA): receives deoxygenated blood from SVC, IVC, and coronary sinus.
- ➡️ Right ventricle (RV): pumps blood through the pulmonary valve into the pulmonary arteries (low resistance circuit).
- 🫁 Left atrium (LA): receives oxygenated blood via four pulmonary veins.
- 💪 Left ventricle (LV): thickest wall, pumps blood through the aortic valve into systemic circulation (high resistance circuit).
- Wall thickness & pressure:
- LV generates ~120 mmHg systolic to overcome systemic vascular resistance; RV generates ~25 mmHg (pulmonary circuit).
- Clinical implication: RV fails more readily with acute afterload rise (e.g., massive PE).
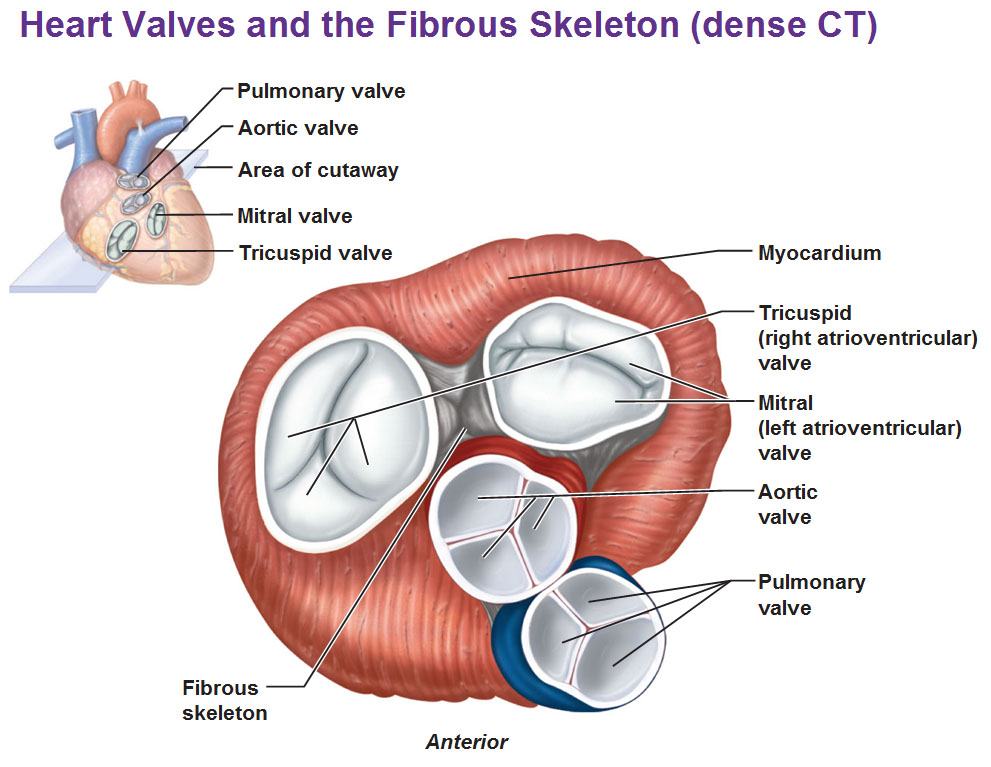
- Cardiac skeleton: fibrous rings support valves, prevent over-distension, and electrically insulate atria from ventricles (so conduction must pass via the AV node/His).
- Valves (one-way doors 🚪):
- Atrioventricular (AV): tricuspid (RA→RV), mitral (LA→LV).
- Semilunar: pulmonary (RV→PA), aortic (LV→aorta).
- Chordae + papillary muscles: prevent AV valve prolapse during systole (do not open/close valves actively; they stabilise leaflets).
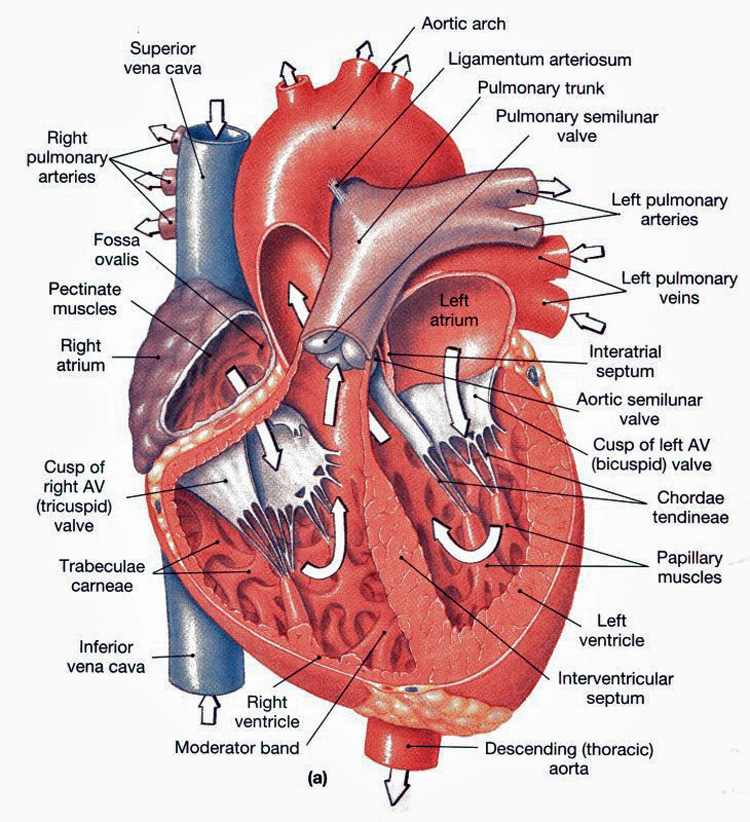
📐 Heart Borders, Surfaces & Key Relations
- Right border: right atrium
- Left border: left ventricle (plus left atrial appendage)
- Inferior (diaphragmatic) surface: mainly LV + part RV
- Anterior (sternocostal) surface: mainly RV
- Base (posterior): mainly LA (important in TOE/mitral disease; close to oesophagus)
📌 Exam Tip: LA enlargement can compress the oesophagus (dysphagia) and, via enlarged pulmonary artery/LA complex, contribute to left recurrent laryngeal nerve palsy (Ortner’s syndrome) causing hoarseness.
🫁 Pericardium (Why it matters clinically)
- Fibrous pericardium: tough outer layer, limits acute distension and anchors heart.
- Serous pericardium: parietal + visceral (epicardium) with lubricating fluid in between.
- Pericardial effusion/tamponade: small rapid effusions can be catastrophic (non-compliant fibrous sac) → impaired diastolic filling → obstructive shock physiology.
🩸 Coronary Circulation & Myocardial Oxygen Supply
Most myocardial oxygen extraction is already high at rest, so increased demand is met mainly by increasing coronary blood flow. Coronary perfusion occurs predominantly in diastole (especially in the LV) because systolic contraction compresses intramyocardial vessels.
- Left main coronary artery → splits into:
- LAD: anterior LV wall + anterior septum (often the “widowmaker” territory).
- Circumflex (LCx): lateral LV wall.
- Right coronary artery (RCA): RV + inferior LV (in right-dominant hearts) + often supplies SA/AV node.
- Dominance: defined by which artery supplies the PDA (posterior descending artery). Right-dominant ~70% (commonly taught).
📌 Exam Tip: Tachycardia shortens diastole → reduced coronary filling time → can precipitate angina, especially with fixed coronary stenoses or LVH.
🔄 The Cardiac Cycle (Mechanical Physiology)
- Systole: isovolumetric contraction → ventricular ejection.
- Diastole: isovolumetric relaxation → rapid passive filling → diastasis → atrial systole (“atrial kick”).
💡 Linking sounds to valves: S1 = closure of mitral/tricuspid (start systole). S2 = closure of aortic/pulmonary (end systole). S3 = rapid filling (can be normal in youth; pathological in volume overload/HF). S4 = atrial contraction into stiff ventricle (LVH/AS/ischaemia).
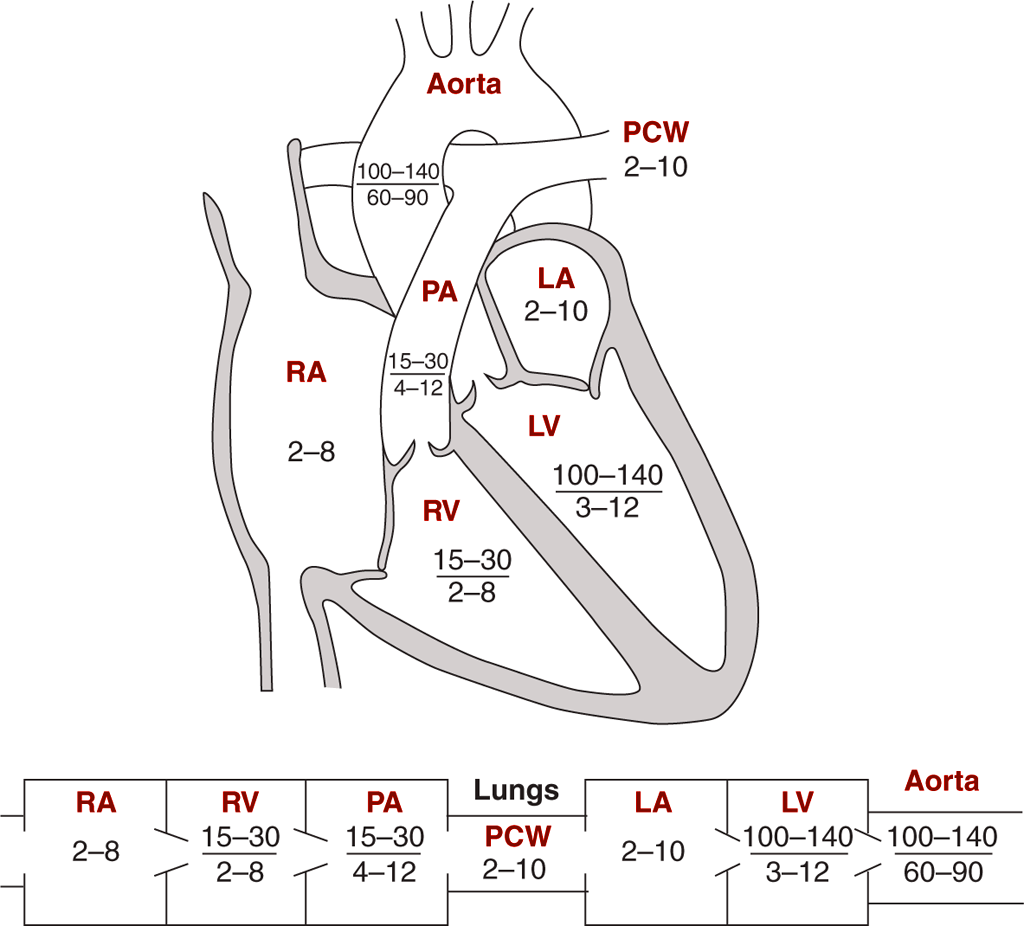
📊 Pressure–Volume (PV) Concepts (High yield)
- Preload: end-diastolic volume (venous return) → stretches sarcomeres.
- Afterload: pressure/resistance ventricle must eject against (aortic pressure/SVR).
- Contractility: intrinsic strength independent of preload (sympathetics/catecholamines ↑).
- Stroke work: area within PV loop; increases with higher afterload and higher stroke volume.
📈 Frank–Starling Law
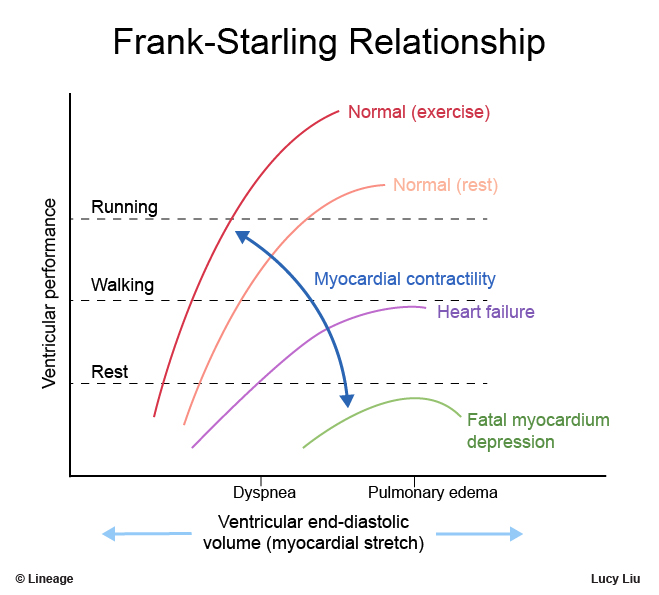
Within physiological limits, ↑ preload → ↑ stroke volume because optimal actin–myosin overlap increases force. In heart failure the curve flattens: extra preload yields little extra output and instead raises filling pressures → pulmonary congestion/oedema.
⚡ Cardiac Electrophysiology: Conduction System
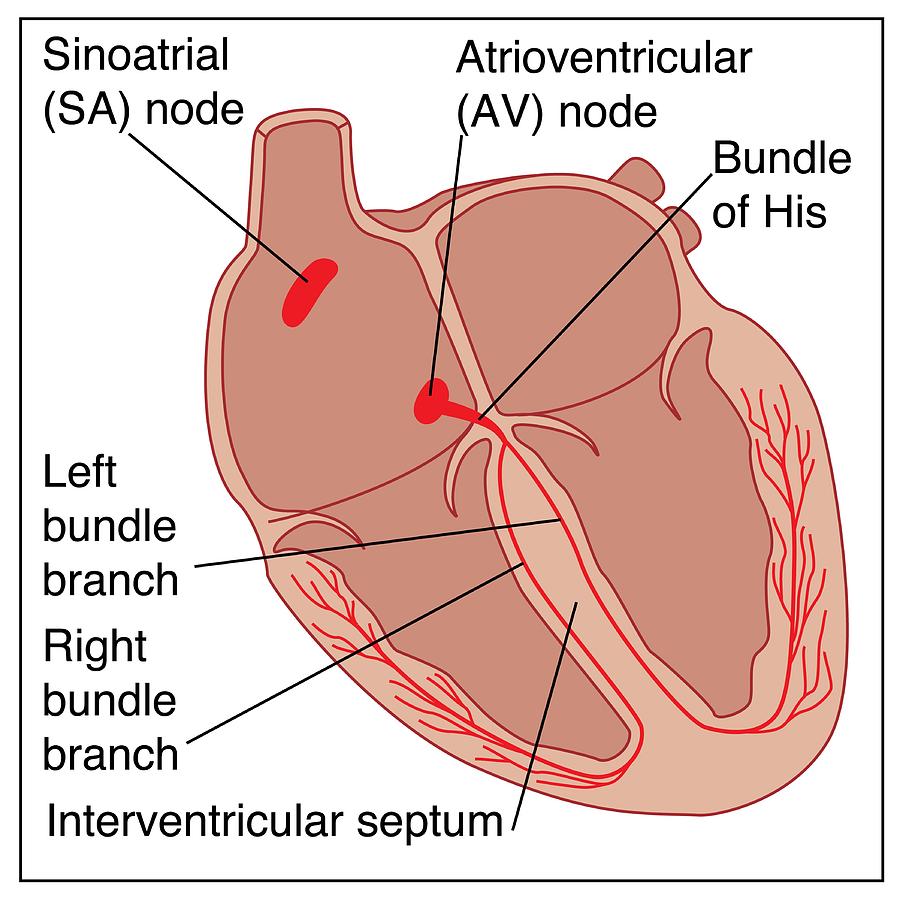
- SA node (dominant pacemaker): typically 60–100 bpm intrinsic.
- Atrial conduction (including Bachmann’s bundle to LA): coordinated atrial depolarisation.
- AV node: slow conduction + physiological delay (~0.1 s) to allow ventricular filling; “gatekeeper” of ventricular rate in atrial arrhythmias.
- His bundle → right/left bundle branches → Purkinje fibres: rapid conduction enabling synchronous ventricular activation.
📌 Exam Tip: Complete heart block = failure of atrial impulses to conduct through AV node/His system → ventricular escape rhythm (often 20–40 bpm) with AV dissociation. Inferior MI (often RCA territory) can cause AV nodal block.
🧠 How the ECG Maps to Physiology
- P wave: atrial depolarisation (atrial contraction follows).
- PR interval: AV nodal delay + His conduction (prolonged in 1st-degree AV block).
- QRS: ventricular depolarisation (mechanical systole begins shortly after).
- ST segment: plateau phase (isoelectric when myocardium uniformly depolarised).
- T wave: ventricular repolarisation.
- QT interval: total time of ventricular depolarisation + repolarisation (prolongation increases torsades risk).
📊 Action Potentials: Pacemaker vs Ventricular Myocyte
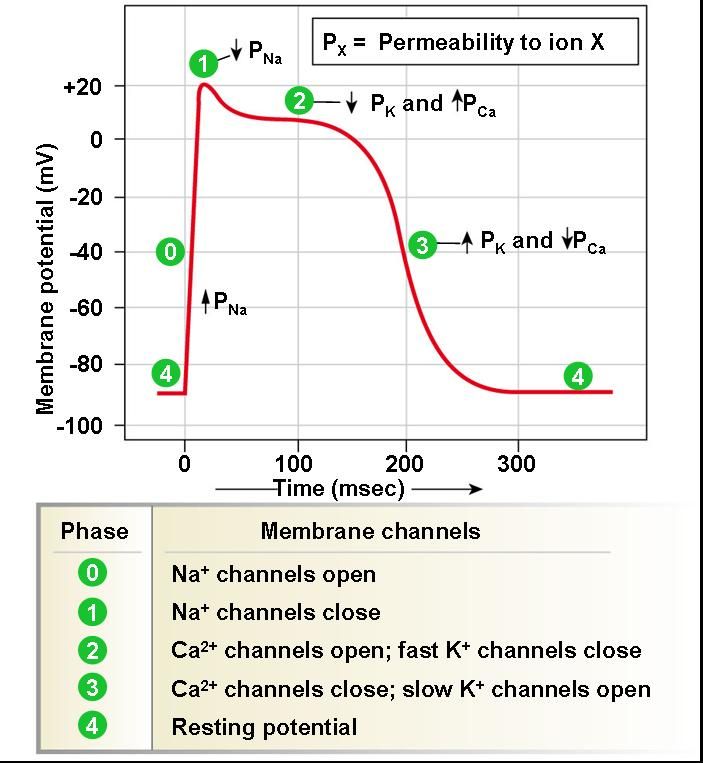
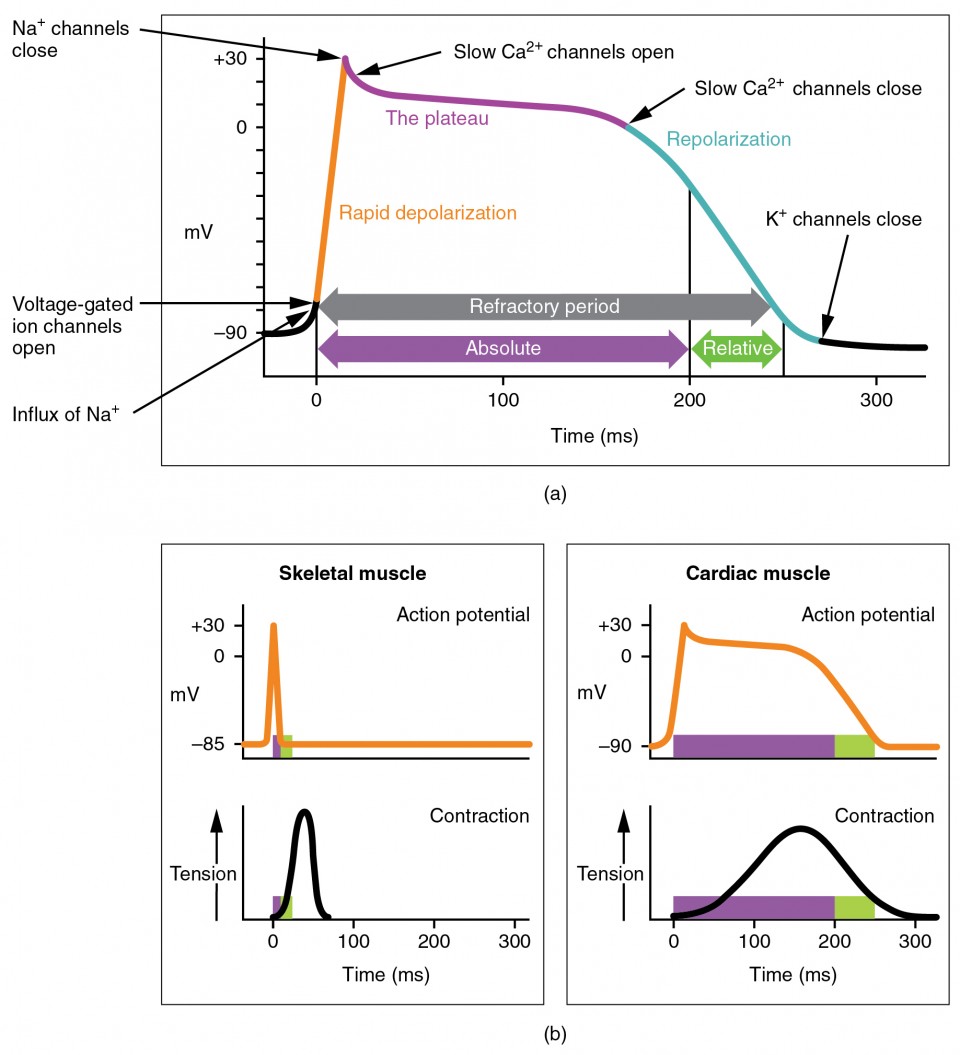
There are two “templates” of cardiac action potential: nodal (pacemaker) cells that spontaneously depolarise, and fast-response atrial/ventricular myocytes that produce a rapid upstroke and a prolonged plateau. The plateau (phase 2) is key: it prolongs the refractory period and prevents tetany, ensuring one coordinated contraction per beat.
- Pacemaker cells (SA/AV node):
- No true resting potential; they slowly drift upward (diastolic depolarisation).
- Phase 4: “funny” current (If, mainly Na⁺ inward) + reduced K⁺ efflux; Ca²⁺ channels contribute late.
- Phase 0: depolarisation mainly via L-type Ca²⁺ influx (not fast Na⁺).
- Phase 3: repolarisation via K⁺ efflux.
- Autonomics: β₁ stimulation increases If and Ca²⁺ currents → steeper phase 4 slope (faster HR); vagal M₂ increases K⁺ conductance and reduces cAMP → slower HR and AV conduction.
- Ventricular myocytes (fast-response):
- Phase 4: stable resting potential ~ −90 mV (high K⁺ permeability via IK1).
- Phase 0: rapid depolarisation via fast Na⁺ channels.
- Phase 1: brief K⁺ out (transient outward current).
- Phase 2 (plateau): Ca²⁺ influx (L-type) balanced by K⁺ efflux → sustained depolarisation.
- Phase 3: repolarisation via delayed rectifier K⁺ currents.
- Refractory period: prolonged-prevents re-entry during normal conduction and stops tetany.
🧲 Resting Membrane Potential & Ion Gradients (Clinically relevant)
The resting membrane potential reflects primarily K⁺ permeability (K⁺ leak channels) and the maintenance of ion gradients by the Na⁺/K⁺-ATPase. Disturbances in extracellular K⁺ shift membrane potential and change excitability-one reason potassium disorders are so arrhythmogenic.
| Ion | Inside (mM) | Outside (mM) | Why it matters |
|---|---|---|---|
| Na⁺ | 10–15 | ~145 | Fast upstroke in myocytes; drives depolarisation and transporters. |
| K⁺ | ~140 | ~4 | Main determinant of resting potential and repolarisation. |
| Cl⁻ | 4–20 | ~110 | Contributes to membrane stability; varies by tissue. |
| Ca²⁺ | ~0.0001 | ~1.8 | Plateau + excitation–contraction coupling; triggers SR release. |
💥 Excitation–Contraction Coupling (Electrical → Mechanical)
When the ventricular action potential reaches the T-tubules, L-type Ca²⁺ channels open and a small Ca²⁺ influx triggers a much larger Ca²⁺ release from the sarcoplasmic reticulum via ryanodine receptors (“calcium-induced calcium release”). Ca²⁺ binds troponin C, enabling actin–myosin cross-bridge cycling and contraction. Relaxation requires energy: Ca²⁺ is pumped back into SR by SERCA and extruded by the Na⁺/Ca²⁺ exchanger.
- Why β₁ agonism increases contractility: ↑ cAMP/PKA → ↑ L-type Ca²⁺ entry + ↑ SR Ca²⁺ loading + faster Ca²⁺ reuptake (stronger + quicker contractions).
- Why ischaemia causes arrhythmias: ATP depletion → pump failure, K⁺ leak, acidosis, heterogeneity of conduction/refractoriness → re-entry risk.
🔁 Arrhythmia Mechanisms (Conceptual framework)
- Abnormal automaticity: ectopic pacemaker activity (e.g., atrial tachycardia).
- Triggered activity: afterdepolarisations:
- EADs (early) with prolonged QT → torsades predisposition.
- DADs (delayed) often with Ca²⁺ overload (e.g., digoxin toxicity, catecholamine excess).
- Re-entry: circuit requires unidirectional block + slow conduction + excitable gap (classic in AVNRT, atrial flutter, VT post-MI scar).
🧠 Autonomic & Hormonal Regulation
- Sympathetic (β₁): ↑ HR (chronotropy), ↑ AV conduction (dromotropy), ↑ contractility (inotropy), ↑ relaxation rate (lusitropy).
- Parasympathetic (vagus, M₂): ↓ SA rate, ↓ AV conduction; strongest effect on atria and AV node.
- RAAS: increases afterload (vasoconstriction) and preload (salt/water retention) → maladaptive in chronic HF.
- ANP/BNP: released with stretch → natriuresis/vasodilation; BNP is a useful biomarker in heart failure pathways.
🏃 Exercise Physiology
- Cardiac output rises via ↑ HR and ↑ stroke volume (enhanced venous return + sympathetic inotropy), commonly reaching ~20–25 L/min in healthy adults.
- Coronary flow increases to match myocardial oxygen demand; failure of this supply–demand match causes exertional angina.
- Redistribution: blood flow shifts toward skeletal muscle and skin (thermoregulation) with maintained cerebral perfusion.
🩺 Clinical Correlations (Make physiology exam-ready)
- Heart failure: reduced effective output triggers neurohormonal activation (SNS/RAAS) → short-term compensation but long-term remodelling and congestion.
- Atrial fibrillation: loss of atrial systole can reduce LV filling (especially in diastolic dysfunction/AS) → breathlessness; irregularly irregular pulse.
- Inferior MI: can cause bradycardia/AV block (AV node often RCA-supplied) and RV infarction (preload sensitive).
- Valvular disease:
- Aortic stenosis: pressure overload → LVH, heaving apex, angina/syncope/HF physiology.
- Mitral regurgitation: volume overload → LA dilatation, AF risk, displaced apex if LV dilates.
📌 Exam Tip: S3 suggests volume overload (HF/dilated cardiomyopathy). S4 suggests a stiff ventricle (LVH, aortic stenosis, ischaemia) and disappears in AF (no atrial contraction).
📝 Summary
The heart’s performance depends on anatomy (chambers/valves/coronaries), mechanics (cardiac cycle, preload/afterload/contractility), and electrophysiology (SA→AV→His-Purkinje conduction and action potentials). The ventricular plateau phase couples electrical activation to contraction and creates a long refractory period that prevents tetany and limits re-entry in normal tissue. Clinically, many symptoms and ECG findings are best understood through these principles: tachycardia reduces diastolic coronary filling, potassium disorders alter excitability, and ischaemia creates conduction heterogeneity that predisposes to arrhythmias.
Categories
- A Level
- About
- Acute Medicine
- Anaesthetics and Critical Care
- Anatomy
- Anatomy and Physiology
- Biochemistry
- Book
- Cardiology
- Collections
- CompSci
- Crib Sheets
- Dental
- Dermatology
- Differentials
- Drugs
- ENT
- Education
- Electrocardiogram
- Embryology
- Emergency Medicine
- Endocrinology
- Ethics
- Foundation Doctors
- GCSE
- Gastroenterology
- General Practice
- Genetics
- Geriatric Medicine
- Guidelines
- Gynaecology
- Haematology
- Hepatology
- Immunology
- Infectious Diseases
- Infographic
- Investigations
- Lists
- MRCP
- Mandatory Training
- Medical Students
- Microbiology
- Nephrology
- Neu
- Neurology
- Neurosurgery
- Nutrition
- OSCE
- OSCEs
- Obstetrics
- Obstetrics Gynaecology
- Oncology
- Ophthalmology
- Oral Medicine and Dentistry
- Orthopaedics
- Paediatrics
- Palliative
- Pathology
- Pharmacology
- Physiology
- Procedures
- Psychiatry
- Public Health
- Radiology
- Renal
- Respiratory
- Resuscitation
- Revision
- Rheumatology
- Statistics and Research
- Stroke
- Surgery
- Toxicology
- Trauma and Orthopaedics
- USMLE
- Urology
- Vascular Surgery
